Because it is bullshit. That's why. Use this curve to calculate the slope and then get back to me.I really don't care what you call me.Well... you did lie about what we concluded, dipshit. If you don't want to be called a dipshit, stop lying.
Let me refer you back my post #179 in the thread Arctic-Heat,
Why don't you tell me why you think the very slow glacial rise of .0005 deg/yr sped up by a factor of 20 to .01 deg/yr in the last few decades.Wuwei said:"But when you look at the far right sections, and at the x and y axis legends, you see the top graph increases about 1 degree in 100 years and the bottom graph (where you circled) increases about 10 degrees in 20,000 years. With that you can calculate the slopes.
If you do the arithmetic the current rise is .01 degrees per year
The glacial rise is about .0005 degrees per year.
That doesn't look the same anymore does it. Are you following this? Have you come across the definition of "slope" in any of your classes? If not you can find a mathematical definition of slope in thefreedictionary.com"
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
Style variation
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
The pseudo science of man-made global warming...
- Thread starter Rustic
- Start date
Wuwei
Gold Member
- Apr 18, 2015
- 5,342
- 1,178
- 255
Of course the two graphs have the same endpoint. They were made that way. I don't know why you get so excited about that. That is of trivial importance.Look at the AGT's of both curves and then get back to me with your bullshit rejection of data.
Your problem is that you were saying the circle in the lower graph encompassed the blue part of the upper graph. That is simply not the case if you look at the hugely different time scales of the two graphs. The circle was way too wide.
Wuwei
Gold Member
- Apr 18, 2015
- 5,342
- 1,178
- 255
I did that twice.Because it is bullshit. That's why. Use this curve to calculate the slope and then get back to me.
SSDD
Gold Member
- Nov 6, 2012
- 16,672
- 1,968
- 280
That is only your opinion. All your sentences are flat wrong except, "objects radiate in all directions according to their temperature."
Sorry guy, but it is not my opinion...and it is stated by the Stefan-Boltzmann law...you have a completely distorted understanding of how, and where energy moves to...
This Equation
Take the black body out of the vacuum and put it in the presence of other matter and you must use this equation
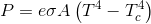
I have already been around with ian on this because he had the same mistaken understanding of the SB law as you....so I took the time to email a few top shelf physicists to ask the simple question and every one that responded answered that yes, the first equation describes a perfect black body in a perfect vacuum radiating according to its temperature...and the second equation described a black body not in a vacuum and therefore radiating according to the difference between its own temperature and the temperature of its surroundings....
You guys like to pretend that you are the smartest fellows in the room, but in order for you to have such a flawed understanding of a perfectly simple equation that elegantly, and simply describes the physical law...clearly you don't have the grasp of the basic science that you believe you do.
I'm talking about radiation physics and you digress into frying pans, marbles, and rust.
You think frying pans, marbles, and rust aren't physics?....you think the laws of physics apply differently to molecules of CO2 than they do to frying pans, marbles and rust?...if so, once again, you prove beyond any doubt that you simply do not have the understanding of physics that you believe that you do and the reason that you have fallen for the alarmist sham of AGW becomes more clear all the time.
This is where you misunderstand entropy. Your mistake is that you are only focusing on the radiation of the colder body to the hotter body. Don't forget that the hotter body will always emit more radiation to the colder body than vice versa. That radiation imbalance means the hotter body cools and the cooler body heats, and entropy increases.
Sorry, but that is where your understanding breaks down...you are under the impression that all matter radiates in every direction according to its temperature....the second expression of the SB law says that is not true...if a radiator is in the presence of another object and it radiates according to the difference between its own temperature and the temperature of its surroundings, then it is not radiating in all directions according to its temperature...
Your whole belief in net energy exchange is faith...not observation....net energy transfer is an artifact of an unobservable, unmeasurable, untestable mathematical model...and in order to be proven, we will need to actually see into the realm of single atom interactions and we aren't even close to that point today...and probably won't be much closer in 100 years.
Again, radiation exchange doesn't violate entropy when it's thermal black body radiation. There is nothing in the definition of entropy that prevents that imbalance of two way radiation.
I suppose nothing in your flawed understanding prevents it, but in reality, if you could read a simple equation, you would see that the very laws of physics prevent it.
SSDD
Gold Member
- Nov 6, 2012
- 16,672
- 1,968
- 280
So now... my final question to you is.... can you be a bigger dipshit than you already are? I hope you are not this incompetent and dishonest in your day job. You wouldn't happen to be a climate scientist, would you? That would explain your lack of competence and honesty.C'mon Ding. We went through that all before. Did you forget that what you circled is actually 20,000 years, not 1,500. Just look at the scale definition at the bottom of the time axis.The last 1500 years is on the graph, Einstein.
You can't blame him...crick taught him to read a graph.
SSDD
Gold Member
- Nov 6, 2012
- 16,672
- 1,968
- 280
So after first admitting you did imply she should stop breathing, but that it was okay because you did it nicely, you've now spun completely about, flipflopping totally, and are declaring you did no such thing.
Liberals are so stupid....are you unable to differentiate between asking someone if they have the courage of their convictions....and to act upon those convictions and simply shooting someone because they don't agree with you? Are you really that stupid hairball?
Never mind, look who I am talking to...of course you are that stupid.
Cool, can you post the graph with the lines you drew?I did that twice.Because it is bullshit. That's why. Use this curve to calculate the slope and then get back to me.
Yeah, no. I never said that. I said the last 1500 years are included in the graph, not that the circle was the 1500 years. I was showing the area that you could find them in.Of course the two graphs have the same endpoint. They were made that way. I don't know why you get so excited about that. That is of trivial importance.Look at the AGT's of both curves and then get back to me with your bullshit rejection of data.
Your problem is that you were saying the circle in the lower graph encompassed the blue part of the upper graph. That is simply not the case if you look at the hugely different time scales of the two graphs. The circle was way too wide.
Wuwei
Gold Member
- Apr 18, 2015
- 5,342
- 1,178
- 255
Sorry guy, but it is not my opinion...
Yes, it's only your opinion. It does not represent science.
You think frying pans, marbles, and rust aren't physics?...etc....
Yes physics. No, not radiation physics.
....so I took the time to email a few top shelf physicists to ask the simple question...
...
Sorry, but that is where your understanding breaks down...you are under the impression that all matter radiates in every direction according to its temperature....the second expression of the SB law says that is not true...i
Did you ask them if it was radiation exchange or net radiation, and not one way radiation? If so they would have answered this way:
http://spie.org/publications/optipe...t/tt48/tt48_154_kirchhoffs_law_and_emissivity
Gustav Robert Kirchhoff (1824–1887) stated in 1860 that “at thermal equilibrium, the power radiated by an object must be equal to the power absorbed.”
https://pediaview.com/openpedia/Radiative_equilibrium
In physics, radiative equilibrium is the condition where a steady state system is in dynamic equilibrium, with equal incoming and outgoing radiative heat flux
Thermal equilibrium • Wikipedia
One form of thermal equilibrium is radiative exchange equilibrium. Two bodies, each with its own uniform temperature, in solely radiative connection, no matter how far apart, or what partially obstructive, reflective, or refractive, obstacles lie in their path of radiative exchange, not moving relative to one another, will exchange thermal radiation, in net the hotter transferring energy to the cooler, and will exchange equal and opposite amounts just when they are at the same temperature.
"http://everything.explained.today/Kirchhoff's_law_of_thermal_radiation/"s_law_of_thermal_radiation/
Kirchhoff's law is that for an arbitrary body emitting and absorbing thermal radiation in thermodynamic equilibrium, the emissivity is equal to the absorptivity.
<<<http://bado-shanai.net/Map of Physics/mopKirchhoffslaw.htm>>>
Imagine a large body that has a deep cavity dug into it. Imagine further that we keep that body at some absolute temperature T and that we have put a small body at a different temperature into the cavity. If the small body has the higher temperature, then it will radiate heat faster than it absorbs heat so that there will be a net flow of heat from the hotter body to the colder body. Eventually the system will come to thermal equilibrium; that is, both bodies will have the same temperature and the small body will emit heat as fast as it absorbs heat.
This is what Max Planck said in 1914.
http://www.gutenberg.org/files/40030/40030-pdf.pdf
Page 31: The energy emitted and the energy absorbed in the state of thermodynamic equilibrium are equal, not only for the entire radiation of the whole spectrum, but also for each monochromatic radiation.
Page 50: "...it is evident that, when thermodynamic equilibrium exists, any two bodies or elements of bodies selected at random exchange by radiation equal amounts of heat with each other..."
---------
If you want more examples, google radiation exchange between black bodies and you will get 1.2 million hits
It is not my understanding it is the understanding of all of physicists. You are essentially saying all physicists have a flawed understanding.I suppose nothing in your flawed understanding prevents it, but in reality, if you could read a simple equation, you would see that the very laws of physics prevent it.
You still didn't give any concept or principle or source that says entropy does not allow two way radiation. You simply can't. Modern science says you are wrong.
Wuwei
Gold Member
- Apr 18, 2015
- 5,342
- 1,178
- 255
Right - the last two pixels of the graph. If that's what you really meant, it would have been clearer if you used an arrow rather than a large circle.Yeah, no. I never said that. I said the last 1500 years are included in the graph, not that the circle was the 1500 years. I was showing the area that you could find them in.
Look at my earlier post where I gave you the end points of the lines in terms of deltas, you can use the same ones and draw your own graph.Cool, can you post the graph with the lines you drew?
SSDD
Gold Member
- Nov 6, 2012
- 16,672
- 1,968
- 280
Yes physics. No, not radiation physics.
The physics of energy is the physics of energy...to the forces that be, marbles, frying pans, and rust are the same as for CO2...
Did you ask them if it was radiation exchange or net radiation, and not one way radiation? If so they would have answered this way:
No need....the SB equation that describes a radiator in the presence of other matter is an equation describing gross one way energy movement...sorry that you can't read an equation and understand what it is describing.
Describe where you think you see two way, net energy exchange in this equation....
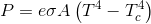
As to what modern science says...sorry, I don't have much respect for modern science...modern science has been telling me for years that salt will cause high blood pressure, and that fat will cause heart disease...and on and on and on with what the consensus thought till it was finally proven by some skeptic that the consensus was full of shit. Every observation ever made supports my position while no observation ever made supports yours...now tell me again about the resonant radio waves that have a temperature.
Old Rocks
Diamond Member
Goddamn, you dumb ****, that does not represent the last 1500 years on that graph. The last 1500 years on that graph would be the last 0.75 % of the distance between 200,000 and 0. And you claim to be an engineer. LOLThe last 1500 years is on the graph, Einstein.Good God, Ding, you have just proven how utterly stupid you are. Very different scales on the two graphs. Were the tow graphs on the same scaling, the upper graph on the climb in temperatures for the last thawing would be a very low slope. About 1 degree per thousand years, compared to the bottom graph of the present warming with is about 1 degree per hundred years. So it is warming about ten times faster than it did coming out of the last glacial period.Of course the current profile matches the temperature profile of the interglacial cycles. Are you smoking crack? The data comes from NASA. The graphs represent global temperature vs time.And yet how do you explain that the present temperature profile matches the previous four interglacial temperature profiles?
But since it doesn't, that's a rather stupid statement on your part.
Even your graphs show it. Anomaly up at +.5C, and still rising. None of the previous cycles passed +.3C.
And by the way, nobody knows what those graphs even represent, since you haven't seen fit to tell us. Are they global? Regional? A single ice core? Being we don't know and you won't tell us, the graphs are meaningless.
Not really. It is all part of a natural cycle that has been occurring for the past 400,000 years. Now do you understand?
I understand the earth should have continued a slow cooldown for at least another 20,000 years, being that was the natural cycle. As a sharp sustained warmup suddenly happened, and we completely skipped thousands of years of global glaciation, climate is certainly not behaving as per the natural cycle.
But please, expound more on your theory. Tell us how completely skipping a natural glaciation cycle is part of the natural cycle. How is the doing the complete opposite of the natural cycle part of the natural cycle?
I'll annotate the graphs to illustrate the stupidity of your argument.
Global Warming : Feature Articles

Old Rocks
Diamond Member
OK, dumb ass, let someone with half a brain spell it out for you. In the last glacial period, the CO2 was 300 ppm for a while. In this interglacial period, the max is about 280 ppm. Until we started dumping GHGs into the atmosphere. At present, the CO2 is 400+ ppm. Fortunately the inertia in the climate is preventing an even more rapid climb in temperatures and sea level. But those will increase, and there will be inevitable surprises as we warm, most of which will not be pleasant.Do the AGT's match for the present, dipshit? Yes they do!C'mon Ding. We went through that all before. Did you forget that what you circled is actually 20,000 years, not 1,500. Just look at the scale definition at the bottom of the time axis.The last 1500 years is on the graph, Einstein.


Old Rocks
Diamond Member
Well, here is one that definitely does just that;Wuwei Still waiting for a link to those studies you claim exist.
Precision tests of QED - Wikipedia
The Wonders of Accuracy: Field Theory and Atomic Spectra
Care to let us know how the two quantum mechanics papers you link to support the theory of AGW.

- Previous article in issue: The impact of new water vapour spectral line parameters on the calculation of atmospheric absorption
- Next article in issue: Polar stratospheric cloud impacts on Antarctic stratospheric heating rates
Volume 127, Issue 575
July 2001 Part A
Pages 1627–1643
Article
Atmospheric absorption of near infrared and visible solar radiation by the hydrogen bonded water dimer
Abstract
Based on the physico-chemical properties of water dimers, their near infrared and visible absorption of solar radiation in the earth's atmosphere is calculated. The calculation uses equilibrium constants determined by statistical mechanics, and a vibrational absorption spectrum determined by a coupled oscillator quantum mechanics model and ab initio quantum chemistry. The resulting total atmospheric absorption was calculated using a line-by-line radiative-transfer model, and depends significantly on the dimer abundance, as well as on the frequency and line width given to vibrational transitions. The best estimate achieved for the possible range of total absorption from 400 nm to 5000 nm by water dimer in the tropics is 1.6–3.3 W m−2. In a global-warming scenario, the increased temperature and water vapour partial pressure result in a nonlinear increase in the absorption of solar radiation by water clusters. Most of the energy from water dimer absorption is deposited in the lower troposphere, particularly in the tropics, tending to make it more convective.
No. I want to see the line that was drawn and I want to see it drawn on the glacial-interglacial plot. How's that?Right - the last two pixels of the graph. If that's what you really meant, it would have been clearer if you used an arrow rather than a large circle.Yeah, no. I never said that. I said the last 1500 years are included in the graph, not that the circle was the 1500 years. I was showing the area that you could find them in.
Look at my earlier post where I gave you the end points of the lines in terms of deltas, you can use the same ones and draw your own graph.Cool, can you post the graph with the lines you drew?
Sorry that is meaningless. We are in an interglacial cycle and our present AGT is still below the temperature last 4 interglacial cycle peaks.OK, dumb ass, let someone with half a brain spell it out for you. In the last glacial period, the CO2 was 300 ppm for a while. In this interglacial period, the max is about 280 ppm. Until we started dumping GHGs into the atmosphere. At present, the CO2 is 400+ ppm. Fortunately the inertia in the climate is preventing an even more rapid climb in temperatures and sea level. But those will increase, and there will be inevitable surprises as we warm, most of which will not be pleasant.Do the AGT's match for the present, dipshit? Yes they do!C'mon Ding. We went through that all before. Did you forget that what you circled is actually 20,000 years, not 1,500. Just look at the scale definition at the bottom of the time axis.The last 1500 years is on the graph, Einstein.



Wuwei
Gold Member
- Apr 18, 2015
- 5,342
- 1,178
- 255
The subject is entropy as it relates to exchange radiation between objects. Falling marbles, etc. are not examples of exchange radiation.The physics of energy is the physics of energy...to the forces that be, marbles, frying pans, and rust are the same as for CO2...
Describe where you think you see two way, net energy exchange in this equation.......To me, and to anyone who can read an equation it states that the power of the radiator is equal to its emissivity times the SB constant times its area times the difference between its own temperature and the temperature of its surroundings....and that is all it says...so describe what you think it says.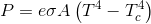
That was already covered in a post about one year ago. All physicists and textbooks agree with this derivation:
This derivation defines equation 3 as a NET radiation exchange and is taught in physics classes. It demonstrates exchange radiation and definitely not one way radiation. Disagree if you want, but you are alone in your opinion.
We are discussing exchange radiation which is important in climate science. Blood pressure, fat, salt, etc. have nothing to do with that topic.As to what modern science says...sorry, I don't have much respect for modern science...modern science has been telling me for years that salt will cause high blood pressure, and that fat will cause heart disease...and on and on and on with what the consensus thought till it was finally proven by some skeptic that the consensus was full of shit. Every observation ever made supports my position while no observation ever made supports yours...now tell me again about the resonant radio waves that have a temperature.
It would be more productive if you could cite scientific sources that say the S-B equation refers to one way radiation. The insert that I gave above denies what you believe. All scientists will disagree with you.
Last edited:
Wuwei
Gold Member
- Apr 18, 2015
- 5,342
- 1,178
- 255
I told you exactly how to draw the lines. I'm not going to wheel-spin if you can't understand how to handle a very simple exercise in finite difference calculus. You would believe the results more if you did it yourself.No. I want to see the line that was drawn and I want to see it drawn on the glacial-interglacial plot. How's that?
Dr Grump
Platinum Member
[Q
And you think that proves exactly what?....that climate science is right even though they don't have the first shred of observed, measured, quantified, empirical evidence supporting the claim that man's CO2 emissions are altering the global climate?....If you do, then you are even more clueless than I first suspected...
Let me ask, do you think there is observed, measured, quantified, empirical evidence in that video supporting the A in the AGW hypothesis?...Give me a time stamp...I will be happy to look and point out to you that it isn't...because no such evidence exists...I do enjoy seeing what passes for actual evidence to you people though...endlessly entertaining and it explains much about why you have been so easily duped.
Can you please post your bona fides? I mean, you seem willing to slam climate scientists. What is your evidence that they are wrong other than you basically saying "I don't believe you". There is so much evidence out there it is unbelievable.
Climate Change: Vital Signs of the Planet: Evidence
Evidence for global warming
Global Warming Science
Old Rocks
Diamond Member
The American Geophysical Union has more climate scientists in it than any other Scientific Society on this planet. This is part of their statement on global warming;
http://sciencepolicy.agu.org/files/2013/07/AGU-Climate-Change-Position-Statement_August-2013.pdf
Human‐Induced Climate Change Requires Urgent Action Humanity is the major influence on the global climate change observed over the past 50 years. Rapid societal responses can significantly lessen negative outcomes. Human activities are changing Earth’s climate. At the global level, atmospheric concentrations of carbon dioxide and other heat‐trapping greenhouse gases have increased sharply since the Industrial Revolution. Fossil fuel burning dominates this increase. Human‐caused increases in greenhouse gases are responsible for most of the observed global average surface warming of roughly 0.8°C (1.5°F) over the past 140 years. Because natural processes cannot quickly remove some of these gases (notably carbon dioxide) from the atmosphere, our past, present, and future emissions will influence the climate system for millennia.
Extensive, independent observations confirm the reality of global warming. These observations show large‐scale increases in air and sea temperatures, sea level, and atmospheric water vapor; they document decreases in the extent of mountain glaciers, snow cover, permafrost, and Arctic sea ice. These changes are broadly consistent with long‐ understood physics and predictions of how the climate system is expected to respond to human‐caused increases in greenhouse gases. The changes are inconsistent with explanations of climate change that rely on known natural influences.
Climate models predict that global temperatures will continue to rise, with the amount of warming primarily determined by the level of emissions. Higher emissions of greenhouse gases will lead to larger warming, and greater risks to society and ecosystems. Some additional warming is unavoidable due to past emissions.
http://sciencepolicy.agu.org/files/2013/07/AGU-Climate-Change-Position-Statement_August-2013.pdf
Human‐Induced Climate Change Requires Urgent Action Humanity is the major influence on the global climate change observed over the past 50 years. Rapid societal responses can significantly lessen negative outcomes. Human activities are changing Earth’s climate. At the global level, atmospheric concentrations of carbon dioxide and other heat‐trapping greenhouse gases have increased sharply since the Industrial Revolution. Fossil fuel burning dominates this increase. Human‐caused increases in greenhouse gases are responsible for most of the observed global average surface warming of roughly 0.8°C (1.5°F) over the past 140 years. Because natural processes cannot quickly remove some of these gases (notably carbon dioxide) from the atmosphere, our past, present, and future emissions will influence the climate system for millennia.
Extensive, independent observations confirm the reality of global warming. These observations show large‐scale increases in air and sea temperatures, sea level, and atmospheric water vapor; they document decreases in the extent of mountain glaciers, snow cover, permafrost, and Arctic sea ice. These changes are broadly consistent with long‐ understood physics and predictions of how the climate system is expected to respond to human‐caused increases in greenhouse gases. The changes are inconsistent with explanations of climate change that rely on known natural influences.
Climate models predict that global temperatures will continue to rise, with the amount of warming primarily determined by the level of emissions. Higher emissions of greenhouse gases will lead to larger warming, and greater risks to society and ecosystems. Some additional warming is unavoidable due to past emissions.
Similar threads
- Replies
- 0
- Views
- 88
- Replies
- 64
- Views
- 2K
- Poll
- Replies
- 132
- Views
- 3K
New Topics
-
Politico poll shows many western nations turning to China, more, as a result of Trump's presidency
- Started by ElmerMudd
- Replies: 8
-
-
Pakistan says it hit militant hideouts in Afghanistan’s Kandahar as fighting shows no letup
- Started by Doc7505
- Replies: 0
-
putin agreed to stop enlisting Kenyans to fight in the Kremlin’s colonial war in Ukraine
- Started by Litwin
- Replies: 0
