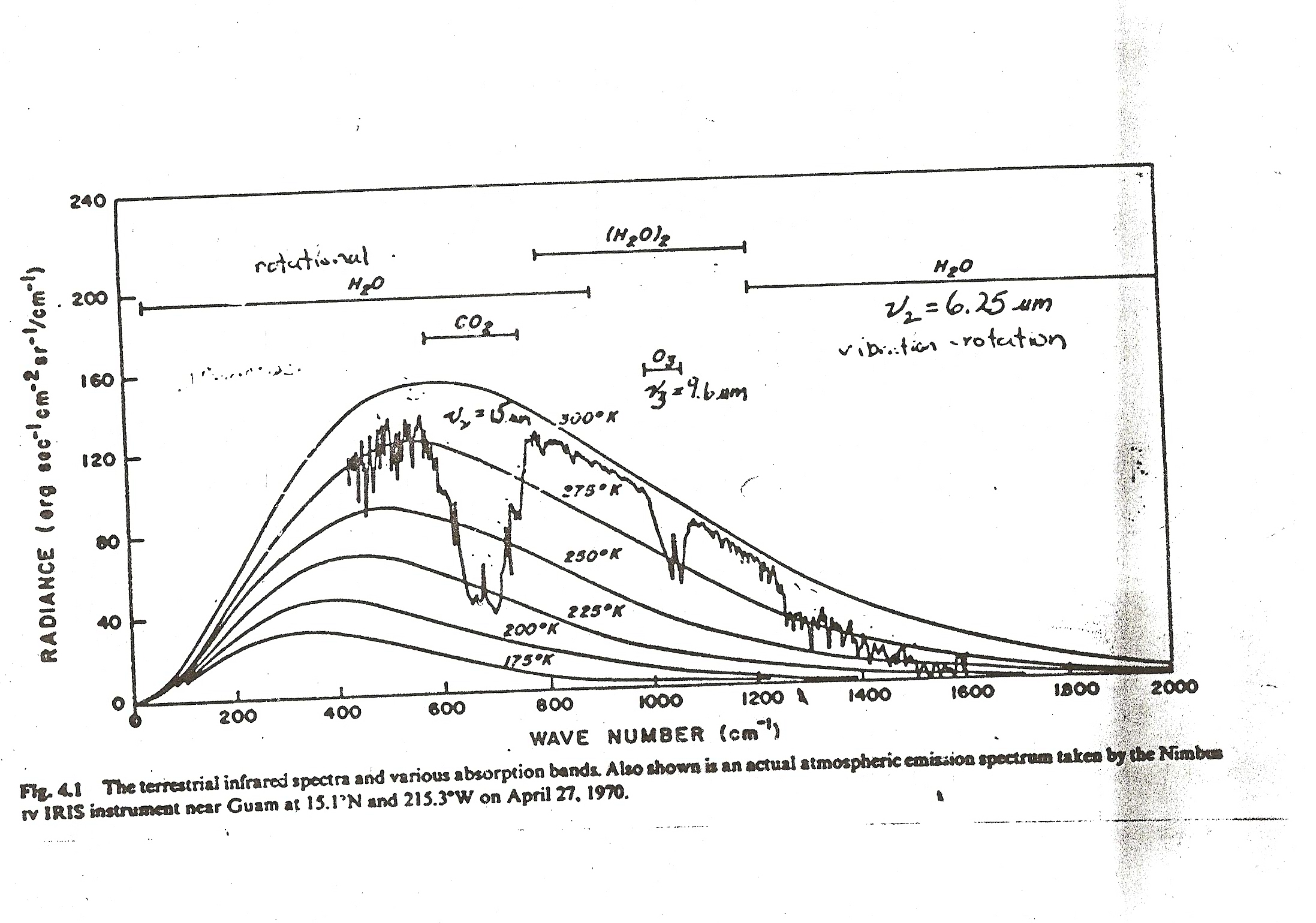
the time between collisions of the various molecules that make up the atmosphere is about a billion times shorter than the time it takes a so called greenhouse gas molecule that has absorbed some IR to actually emit it as radiation
I find the time to emit is roughly 8 nS.
The average velocity of molecules in the atmosphere is 1866 km/hr or 51,883 cm/s. The mean free path is approximately 70 nanometers. That gives a mean time between collisions of 1.35057e-10 seconds or 0.135057 nanoseconds. Thus collisions occur about 60 times as frequently as spontaneous emissions, not billions of times.
I concur that emissions happen about one order of magnitude less often than collisions but certainly more often than SSDDs claim of one in a billion.
Obviously the percentage of excited molecules actually emitting increases the further up into the atmosphere you go, due to less density and fewer collisions.
Email Dr Happer...Most of those guys are easily accessible and more than willing to answer questions....I certainly would be more likely to accept a statement by someone of the stature of Dr Happer than any of our local warmer wackos...including yourself.
I have a problem with Happer's comments in as much as he is giving the odds of an individual CO2 molecule absorbing and then emitting a photon. The vast majority of excited CO2 molecules become excited by collision not absorption.
Let us turn to a slightly different area. You have claimed repeatedly that "absorption and emission does not equal warming". How do you reconcile that statement with Happer's comments that 99.99% of the radiation absorbed by CO2 does get added to the total energy of the atmosphere, some of which would be expressed as kinetic energy, measured as temperature?
Energy absorbed by CO2 does not get "added" it is just part of the energy radiating from the earth...Just because CO2 absorbs a bit of IR does not mean that it gets "added" to the total energy...it was already there....why would it get added?
the time between collisions of the various molecules that make up the atmosphere is about a billion times shorter than the time it takes a so called greenhouse gas molecule that has absorbed some IR to actually emit it as radiation
I find the time to emit is roughly 8 nS.
The average velocity of molecules in the atmosphere is 1866 km/hr or 51,883 cm/s. The mean free path is approximately 70 nanometers. That gives a mean time between collisions of 1.35057e-10 seconds or 0.135057 nanoseconds. Thus collisions occur about 60 times as frequently as spontaneous emissions, not billions of times.
I concur that emissions happen about one order of magnitude less often than collisions but certainly more often than SSDDs claim of one in a billion.
Obviously the percentage of excited molecules actually emitting increases the further up into the atmosphere you go, due to less density and fewer collisions.
Email Dr Happer...Most of those guys are easily accessible and more than willing to answer questions....I certainly would be more likely to accept a statement by someone of the stature of Dr Happer than any of our local warmer wackos...including yourself.
I have a problem with Happer's comments in as much as he is giving the odds of an individual CO2 molecule absorbing and then emitting a photon. The vast majority of excited CO2 molecules become excited by collision not absorption.
Let us turn to a slightly different area. You have claimed repeatedly that "absorption and emission does not equal warming". How do you reconcile that statement with Happer's comments that 99.99% of the radiation absorbed by CO2 does get added to the total energy of the atmosphere, some of which would be expressed as kinetic energy, measured as temperature?
Energy absorbed by CO2 does not get "added" it is just part of the energy radiating from the earth...Just because CO2 absorbs a bit of IR does not mean that it gets "added" to the total energy...it was already there....why would it get added?
The source of the IR is the surface. The surface is shedding energy by producing radiation. Some of the IR escapes directly to space.
But some of this IR radiation energy is absorbed by CO2 in the atmosphere (all within the first 10 meters for 15 micron).
This energy has been removed from the surface and added to the atmosphere. My comment explicitly said "added to the total energy of the atmosphere". I further explained that part of this energy would be expressed as kinetic energy (as opposed to potential energy), which by definition means a warmer temperature.
Where are you confused about this simple explanation? Be specific, maybe I can help clear it up.