Folks - people will tell you that you need not be worried because adjuvants aren't currently being used in H1N1 vaccines that are currently approved for distribution. What these people don't understand is that the government is planning to use them once fast-track manufacturing and trials have been completed. The folllowing includes a listing of trials and relevant data. A link is included to most of the studies.
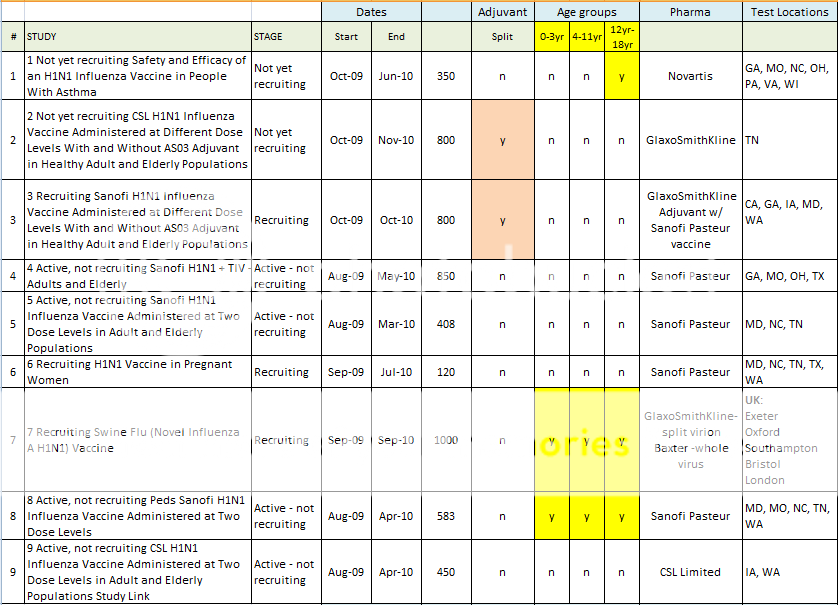
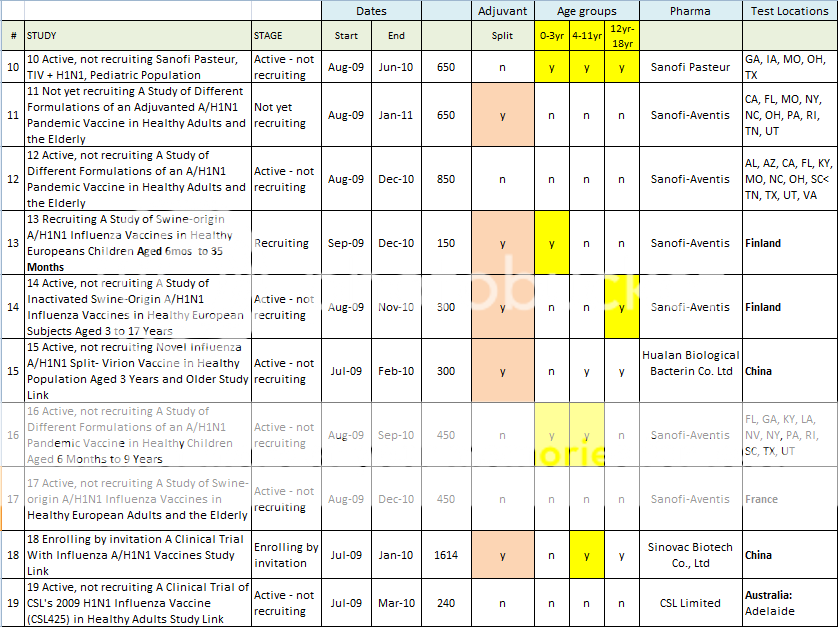
Those vaccines with red highlights include adjuvants. There's a table at the bottom that highlights the use of adjuvants, and that highlights the clinical trials that include experimental vaccination of children from 0-18 years old.
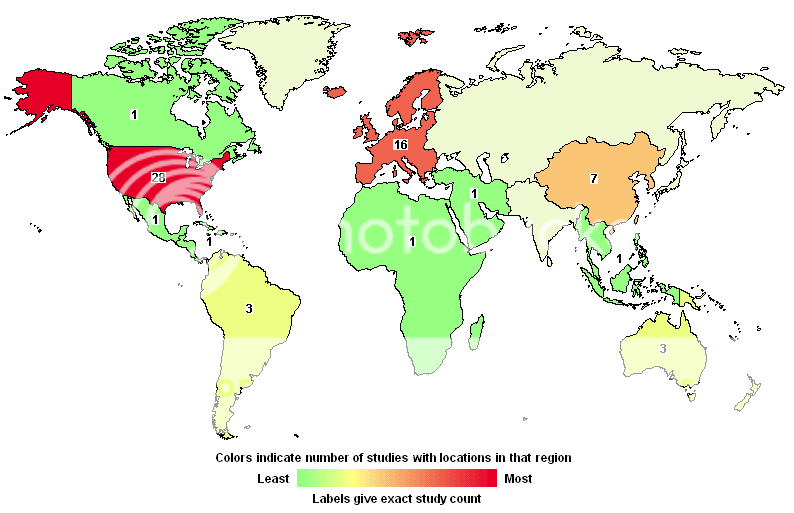
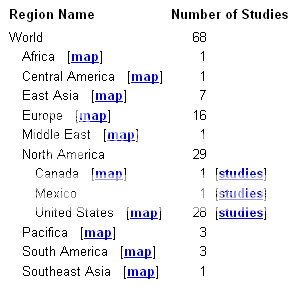
All Current H1N1 Clinical Trials: Index
This lists all current H1N1 clinical trials registered with the US government.
The status of each study will get updated in this index as the trials progress, and I'll post the study results when available.
Status: Not yet Recruiting, Recruiting, Enrolling by Invitation,
Active-recruiting, Active-not recruiting,
Suspended, Completed, Completed (results available)
This map is interactive at the source: Search of: H1N1 - Results on Map - ClinicalTrials.gov
1 Not yet recruiting Safety and Efficacy of an H1N1 Influenza Vaccine in People With Asthma Study Link
Biological: H1N1 vaccine high dose; Biological: H1N1 vaccine low dose
2 Not yet recruiting CSL H1N1 Influenza Vaccine Administered at Different Dose Levels With and Without AS03 Adjuvant in Healthy Adult and Elderly Populations Study Link
Biological: AS03; Biological: Inactivated H1N1 Vaccine
3 Recruiting Sanofi H1N1 Influenza Vaccine Administered at Different Dose Levels With and Without AS03 Adjuvant in Healthy Adult and Elderly Populations Study Link
Biological: AS03; Biological: Inactivated H1N1 Vaccine
4 Active, not recruiting Sanofi H1N1 + TIV - Adults and Elderly Study Link
Biological: Inactivated H1N1 Vaccine; Drug: Placebo; Biological: TIV
5 Active, not recruiting Sanofi H1N1 Influenza Vaccine Administered at Two Dose Levels in Adult and Elderly Populations Study Link
Biological: Inactivated H1N1 Vaccine
6 Recruiting H1N1 Vaccine in Pregnant WomenStudy Link
Biological: Inactivated H1N1 Vaccine
7 Recruiting Swine Flu (Novel Influenza A H1N1) Vaccine Study Study Link
Biological: Baxter Novel Influenza A H1N1 Whole Virus Vaccine;
Biological: GlaxoSmithKline Novel Influenza A H1N1 Split Virion Vaccine
8 Active, not recruiting Peds Sanofi H1N1 Influenza Vaccine Administered at Two Dose Levels Study Link
Biological: Inactivated H1N1 Vaccine
9 Active, not recruiting CSL H1N1 Influenza Vaccine Administered at Two Dose Levels in Adult and Elderly Populations Study Link
Biological: Inactivated H1N1 Vaccine
10 Active, not recruiting Sanofi Pasteur, TIV + H1N1, Pediatric Population Study Link
Biological: Inactivated H1N1 Vaccine;
Biological: Trivalent Inactivated Influenza Vaccine
11 Not yet recruiting A Study of Different Formulations of an Adjuvanted A/H1N1 Pandemic Vaccine in Healthy Adults and the Elderly Study Link
Influenza; Swine-origin A/H1N1 Influenza
Biological: Monovalent Subvirion A/H1N1 influenza vaccine with adjuvant;
Biological: Monovalent Subvirion A/H1N1 influenza vaccine;
Biological: Normal saline solution
12 Active, not recruiting A Study of Different Formulations of an A/H1N1 Pandemic Vaccine in Healthy Adults and the Elderly Study Link
Influenza; Swine-origin A/H1N1 Influenza
Biological: Monovalent Subvirion A/H1N1 Influenza vaccine (7.5 µg of HA);
Biological: Monovalent Subvirion A/H1N1 Influenza vaccine (15 µg of HA);
Biological: Monovalent Subvirion A/H1N1 Influenza vaccine (30 µg of HA);
Biological: Normal saline solution (placebo)
13 Recruiting A Study of Swine-origin A/H1N1 Influenza Vaccines in Healthy Europeans Children Aged 6 to 35 Months Study Link
Influenza; Swine-origin A/H1N1 Influenza
Biological: Swine A/H1N1 influenza vaccine (split virion, inactivated);
Biological: Swine A/H1N1 influenza vaccine (split virion, inactivated + Adjuvant);
Biological: Swine A/H1N1 influenza vaccine (split virion, inactivated + adjuvant)
14 Active, not recruiting A Study of Inactivated Swine-Origin A/H1N1 Influenza Vaccines in Healthy European Subjects Aged 3 to 17 Years Study Link
Influenza; Swine-origin A/H1N1 Influenza
Biological: Swine A/H1N1 influenza vaccine (split virion, inactivated);
Biological: Swine A/H1N1 influenza vaccine (split virion, inactivated + Adjuvant)
15 Active, not recruiting Novel Influenza A/H1N1 Split- Virion Vaccine in Healthy Population Aged 3 Years and Older Study Link
Biological: split-virion, non-adjuvanted H1N1 vaccine of 30 μg;
Biological: Experimental: split-virion, adjuvanted H1N1 vaccine of 7.5 μg;
Biological: Experimental: split-virion, adjuvanted H1N1 vaccine of 15 μg;
Biological: Experimental: split-virion, adjuvanted H1N1 vaccine of 30 μg;
Biological: Experimental: split-virion, non-adjuvanted H1N1 vaccine of 15 μg;
Biological: Placebo Comparator: Placebo control
16 Active, not recruiting A Study of Different Formulations of an A/H1N1 Pandemic Vaccine in Healthy Children Aged 6 Months to 9 Years Study Link
Biological: Monovalent Subvirion A/H1N1 Influenza vaccine;
Biological: Normal saline solution (placebo)
17 Active, not recruiting A Study of Swine-origin A/H1N1 Influenza Vaccines in Healthy European Adults and the Elderly Study Link
Biological: Swine A/H1N1 influenza vaccine (split virion, inactivated)
18 Enrolling by invitation A Clinical Trial With Influenza A/H1N1 Vaccines Study Link
Influenza
Biological: split-virion, adjuvanted H1N1 vaccine of 7.5 μg per dose;
Biological: split-virion, adjuvanted H1N1 vaccine of 15 μg per dose;
Biological: split-virion, non-adjuvanted H1N1 vaccine of 15 μg per dose;
Biological: split-virion, non-adjuvanted H1N1 vaccine of 30 μg per dose;
Biological: whole-virion, adjuvanted H1N1 vaccine of 5 μg per dose;
Biological: whole-virion, adjuvanted H1N1 vaccine of 10 μg per dose;
Biological: placebo control
19 Active, not recruiting A Clinical Trial of CSL's 2009 H1N1 Influenza Vaccine (CSL425) in Healthy Adults Study Link
Biological: CSL's 2009 H1N1 Influenza Vaccine (CSL425)
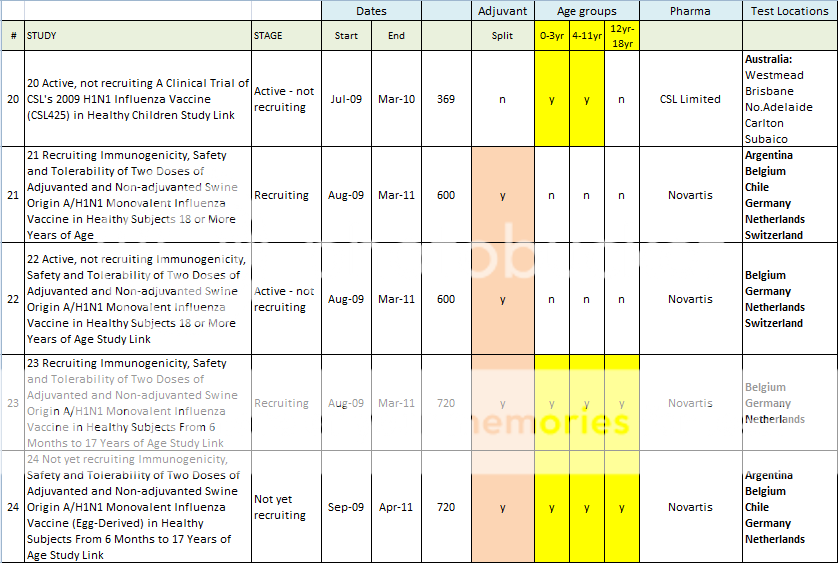
20 Active, not recruiting A Clinical Trial of CSL's 2009 H1N1 Influenza Vaccine (CSL425) in Healthy Children Study Link
Biological: CSL's 2009 H1N1 Influenza Vaccine (CSL425)
21 Recruiting Immunogenicity, Safety and Tolerability of Two Doses of Adjuvanted and Non-adjuvanted Swine Origin A/H1N1 Monovalent Influenza Vaccine in Healthy Subjects 18 or More Years of Age Study Link
Biological: Monovalent A/H1N1 influenza vaccine
22 Active, not recruiting Immunogenicity, Safety and Tolerability of Two Doses of Adjuvanted and Non-adjuvanted Swine Origin A/H1N1 Monovalent Influenza Vaccine in Healthy Subjects 18 or More Years of Age Study Link
Biological: Monovalent A/H1N1 influenza vaccine
23 Recruiting Immunogenicity, Safety and Tolerability of Two Doses of Adjuvanted and Non-adjuvanted Swine Origin A/H1N1 Monovalent Influenza Vaccine in Healthy Subjects From 6 Months to 17 Years of Age Study Link
Biological: Monovalent A/H1N1 influenza vaccine
24 Not yet recruiting Immunogenicity, Safety and Tolerability of Two Doses of Adjuvanted and Non-adjuvanted Swine Origin A/H1N1 Monovalent Influenza Vaccine (Egg-Derived) in Healthy Subjects From 6 Months to 17 Years of Age Study Link
Biological: Monovalent A/H1N1 influenza vaccine
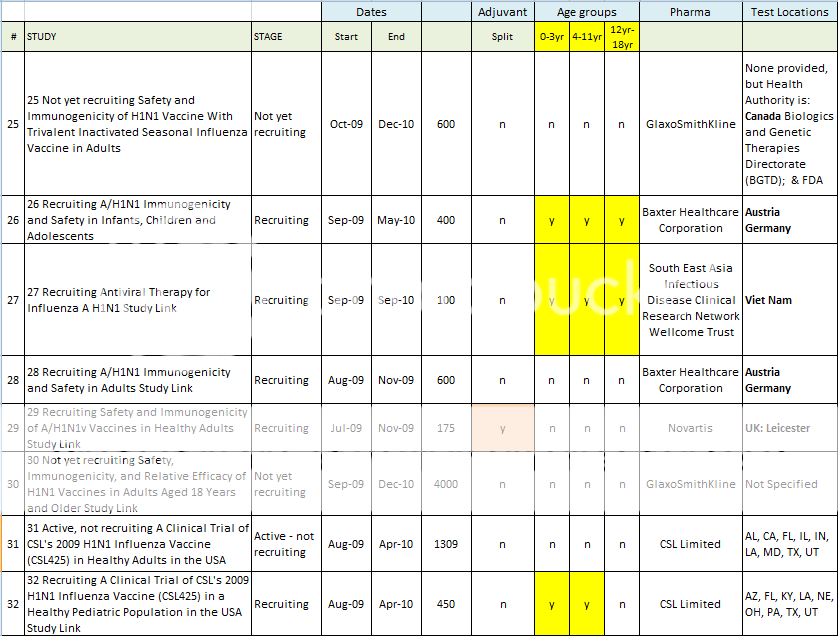
25 Not yet recruiting Safety and Immunogenicity of H1N1 Vaccine With Trivalent Inactivated Seasonal Influenza Vaccine in Adults Study Link
Biological: Seasonal trivalent influenza vaccine (TIV);
Biological: GSK2340274A;
Biological: GSK2340273A;
Biological: Saline placebo
26 Recruiting A/H1N1 Immunogenicity and Safety in Infants, Children and Adolescents Study Link
Biological: H1N1 pandemic influenza vaccine (whole virion, Vero Cell-derived, inactivated)
27 Recruiting Antiviral Therapy for Influenza A H1N1 Study Link
28 Recruiting A/H1N1 Immunogenicity and Safety in Adults Study Link
Biological: H1N1 pandemic influenza vaccine (whole virion, Vero Cell-derived, inactivated)
29 Recruiting Safety and Immunogenicity of A/H1N1v Vaccines in Healthy Adults Study Link
Biological: MF59 H1N1 vaccine;
Biological: Plain H1N1 vaccine
30 Not yet recruiting Safety, Immunogenicity, and Relative Efficacy of H1N1 Vaccines in Adults Aged 18 Years and Older Study Link
Biological: GSK2340274A;
Biological: GSK2340273A
31 Active, not recruiting A Clinical Trial of CSL's 2009 H1N1 Influenza Vaccine (CSL425) in Healthy Adults in the USA Study Link
Biological: CSL's 2009 H1N1 Influenza Vaccine (CSL425);
Biological: Placebo
32 Recruiting A Clinical Trial of CSL's 2009 H1N1 Influenza Vaccine (CSL425) in a Healthy Pediatric Population in the USA Study Link
Biological: CSL's 2009 H1N1 Influenza Vaccine (CSL425); Biological: Placebo
33 Recruiting A Pilot Study for Collection of Anti-Influenza A H1N1 (Swine Flu) Hyper-Immune Plasma Study Link (Note: Although related to H1N1, this is not a vaccine trial).
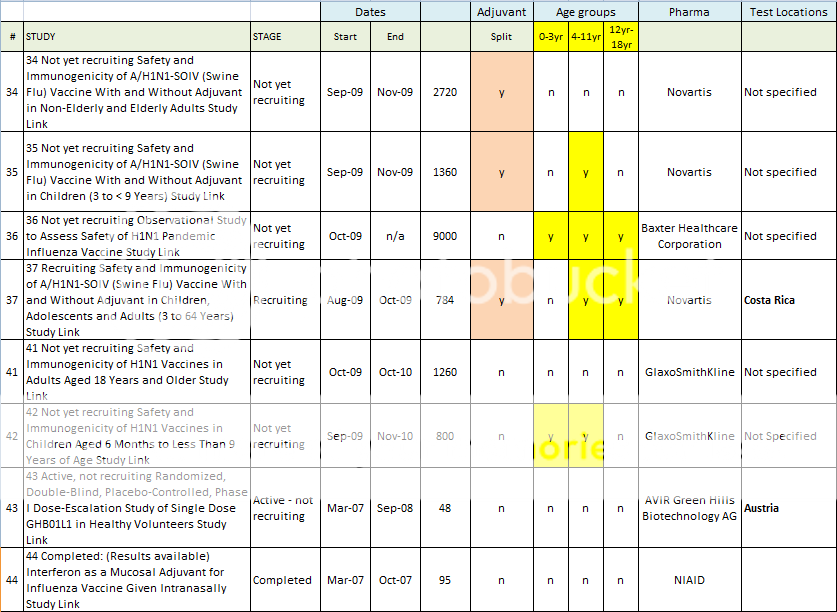
34 Not yet recruiting Safety and Immunogenicity of A/H1N1-SOIV (Swine Flu) Vaccine With and Without Adjuvant in Non-Elderly and Elderly Adults Study Link
Biological: Adjuvanted and un-adjuvanted influenza vaccines
35 Not yet recruiting Safety and Immunogenicity of A/H1N1-SOIV (Swine Flu) Vaccine With and Without Adjuvant in Children (3 to < 9 Years) Study Link
Biological: Adjuvanted and un-adjuvanted influenza vaccines
36 Not yet recruiting Observational Study to Assess Safety of H1N1 Pandemic Influenza Vaccine Study Link
Biological: H1N1 Pandemic Influenza Vaccine (whole virion, Vero Cell-derived, inactivated)
37 Recruiting Safety and Immunogenicity of A/H1N1-SOIV (Swine Flu) Vaccine With and Without Adjuvant in Children, Adolescents and Adults (3 to 64 Years) Study Link
Biological: Adjuvanted and un-adjuvanted influenza vaccines
38 Recruiting Chinese Medicinal Herbs Treatment on Novel Influenza A (H1N1) : Multi-centre, Prospective, Randomized Controlled Study Study Link
(Note: Although related to H1N1, this is not a vaccine trial).
39 Not yet recruiting Chinese Medicinal Herbs Treatment on Novel Influenza A (H1N1)Pneumonia: Multi-centre, Prospective, Randomized Controlled Study Study Link
(Note: Although related to H1N1, this is not a vaccine trial).
40 Not yet recruiting A Clinical Trial Comparing Oseltamivir With Placebo And Zanamivir With Control As The First Line Treatment For Human Swine Influenza (H1N1) Infection During The Pandemic Influenza in Hong Kong Study Link
(Note: Although related to H1N1, this is not a vaccine trial).
41 Not yet recruiting Safety and Immunogenicity of H1N1 Vaccines in Adults Aged 18 Years and Older Study Link
Biological: Saline placebo;
Biological: GSK2340274A;
Biological: GSK2340273A
42 Not yet recruiting Safety and Immunogenicity of H1N1 Vaccines in Children Aged 6 Months to Less Than 9 Years of Age Study Link
43 Active, not recruiting Randomized, Double-Blind, Placebo-Controlled, Phase I Dose-Escalation Study of Single Dose GHB01L1 in Healthy Volunteers Study Link
Biological: GHB01L1
44 Completed: (Results available) Interferon as a Mucosal Adjuvant for Influenza Vaccine Given Intranasally Study Link
Posted here: Login
Biological: Trivalent inactivated influenza virus vaccine (2006-2007 formulation);
Biological: Type 1 interferon Has Results Interferon as a Mucosal Adjuvant for Influenza Vaccine Given Intranasally
Biological: Trivalent inactivated influenza virus vaccine (2006-2007 formulation); Biological: Type 1 interferon
45 Not yet recruiting STIP: Statin Trial for Influenza Patients Study Link
Drug: Rosuvastatin (crestor);
Drug: Placebo
(Note: Not an H1N1 Vaccine trial)
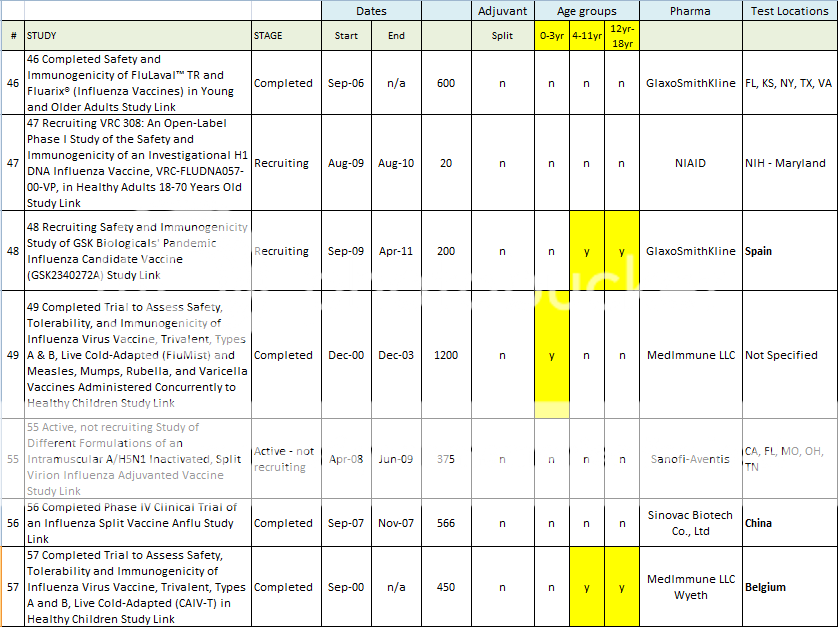
46 Completed Safety and Immunogenicity of FluLaval TR and Fluarix® (Influenza Vaccines) in Young and Older Adults Study Link
Biological: Influenza vaccine (A/H3N2, A/H1N1, and B strains); Biological: Fluarix
Posted here:
Login
47 Recruiting VRC 308: An Open-Label Phase I Study of the Safety and Immunogenicity of an Investigational H1 DNA Influenza Vaccine, VRC-FLUDNA057-00-VP, in Healthy Adults 18-70 Years Old Study Link
Biological: VRC-FLUDNA057-00-VP
48 Recruiting Safety and Immunogenicity Study of GSK Biologicals' Pandemic Influenza Candidate Vaccine (GSK2340272A) Study Link
Biological: Pandemic influenza vaccine GSK2340272A
49 Completed Trial to Assess Safety, Tolerability, and Immunogenicity of Influenza Virus Vaccine, Trivalent, Types A & B, Live Cold-Adapted (FluMist) and Measles, Mumps, Rubella, and Varicella Vaccines Administered Concurrently to Healthy Children Study Link
Biological: FluMist; Other: Placebo
Posted here:
Login
50 Recruiting A Study on Emergence of Resistance With Tamiflu (Oseltamivir) in Patients With Seasonal Influenza Study Link
Drug: Tamiflu (oseltamivir)
(Note: Not an H1N1 vaccine study: they are studying Tamiflu)
51 Suspended Prospective Evaluation of the Fluid Rapid Influenza Test Study Link
Influenza
Device: fluID Rapid Influenza Test
(Note: Not an H1N1 vaccine study: diagnostic study)
52 Recruiting Direct and Indirect Benefits of Influenza Vaccination in Schools and Households Study Link
Biological: Trivalent live attenuated seasonal influenza vaccine;
Biological: Monovalent live attenuated pandemic influenza vaccine;
Biological: Saline
(Note: Not an H1N1 vaccine study: they are studying the symptoms of flu - no meds)
53 Not yet recruiting Mechanisms of Severe Acute Influenza Consortium (MOSAIC) Study Link
54 Recruiting Safety, Immunogenicity Study of GSK Biologicals' Pandemic Influenza Candidate Vaccine (GSK2340272A) Study Link
Biological: Pandemic influenza vaccine GSK2340272A
55 Active, not recruiting Study of Different Formulations of an Intramuscular A/H5N1 Inactivated, Split Virion Influenza Adjuvanted Vaccine Study Link
Biological: Monovalent subvirion H5N1 influenza vaccine;
Biological: Physiological saline
56 Completed Phase IV Clinical Trial of an Influenza Split Vaccine Anflu Study Link
Biological: seasonal split influenza vaccine
57 Completed Trial to Assess Safety, Tolerability and Immunogenicity of Influenza Virus Vaccine, Trivalent, Types A and B, Live Cold-Adapted (CAIV-T) in Healthy Children Study Link
Biological: CAIV-T
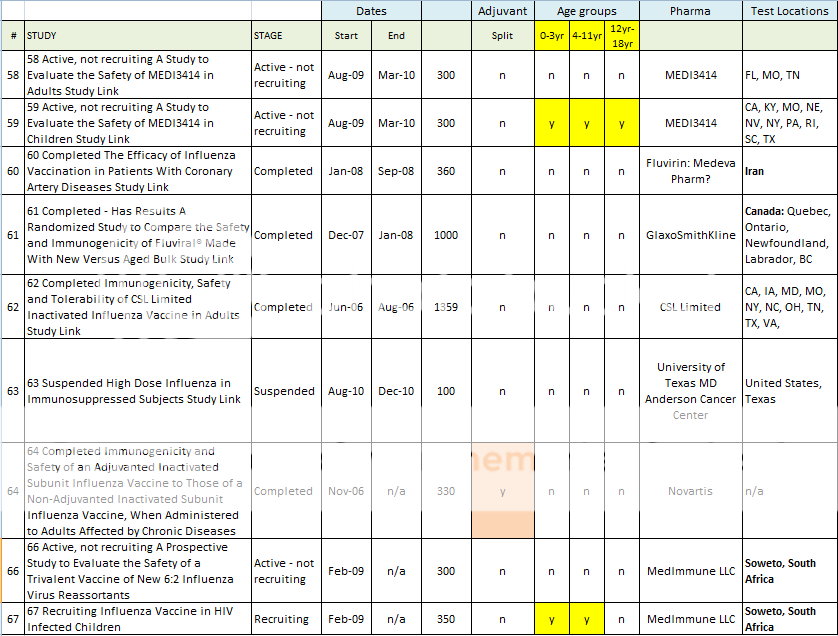
58 Active, not recruiting A Study to Evaluate the Safety of MEDI3414 in Adults Study Link
Biological: MEDI3414; Other: Placebo
59 Active, not recruiting A Study to Evaluate the Safety of MEDI3414 in Children Study Link
Biological: MEDI3414;
Other: Placebo
60 Completed The Efficacy of Influenza Vaccination in Patients With Coronary Artery Diseases Study Link
Coronary Artery Diseases; Myocardial Infarction; Stable Angina
Biological: influenza vaccine;
Biological: placebo for influenza vaccine
Biological: Fluviral
61 Completed - Has Results A Randomized Study to Compare the Safety and Immunogenicity of Fluviral® Made With New Versus Aged Bulk Study Link
Biological: Fluviral
62 Completed Immunogenicity, Safety and Tolerability of CSL Limited Inactivated Influenza Vaccine in Adults Study Link
Influenza
Biological: CSL Trivalent Inactivated Influenza Vaccine - Thimerosal-free;
Biological: CSL Trivalent Inactivated Influenza Vaccine with Thimerosal;
Biological: Placebo with Thimerosal
63 Suspended High Dose Influenza in Immunosuppressed Subjects Study Link
Biological: TIV;
Biological: rHAO Trivalent Influenza Vaccine
(Note: Study on cancer patients w/9x vaccine dosage - suspended; no reasons provided for suspension)
64 Completed Immunogenicity and Safety of an Adjuvanted Inactivated Subunit Influenza Vaccine to Those of a Non-Adjuvanted Inactivated Subunit Influenza Vaccine, When Administered to Adults Affected by Chronic Diseases Study Link
Influenza Disease
Biological: Adjuvanted influenza vaccine
(Note: Study completed: but no results posted, no locations for testing, no end date for the study!)
65 Completed Safety and Immunogenicity Study of Hib-MenCY-TT Vaccine Compared to Licensed Hib Conjugate Vaccine Study Link
(Note: Not an H1N1 Vaccine Trial, although H1N1 is referenced.)
66 Active, not recruiting A Prospective Study to Evaluate the Safety of a Trivalent Vaccine of New 6:2 Influenza Virus Reassortants (on HIV-infected Children) Study Link
Biological: FluMist;
Biological: Placebo
67 Recruiting Influenza Vaccine in HIV Infected Children Study Link
Viral-Confirmed Influenza Illness
Biological: Trivalent sub-unit influenza vaccine;
Biological: Saline, 0.5ml
68 Suspended Comparison of Fluid Rapid Influenza and BinaxNOW Influenza A & B Study Link
(Note: Not an H1N1 Vaccine Trial)
Here is a spreadsheet view of all of the H1N1 Clinical Trials listed in the Index above. Note that I did not include any trials that did not include H1N1 vaccine in testing. (As in tests for Tamiflu, or for injection devices. )
Summary of H1N1 Clinical Trials:
 37 PM by NVIC Administrator | with 0 comments
37 PM by NVIC Administrator | with 0 comments