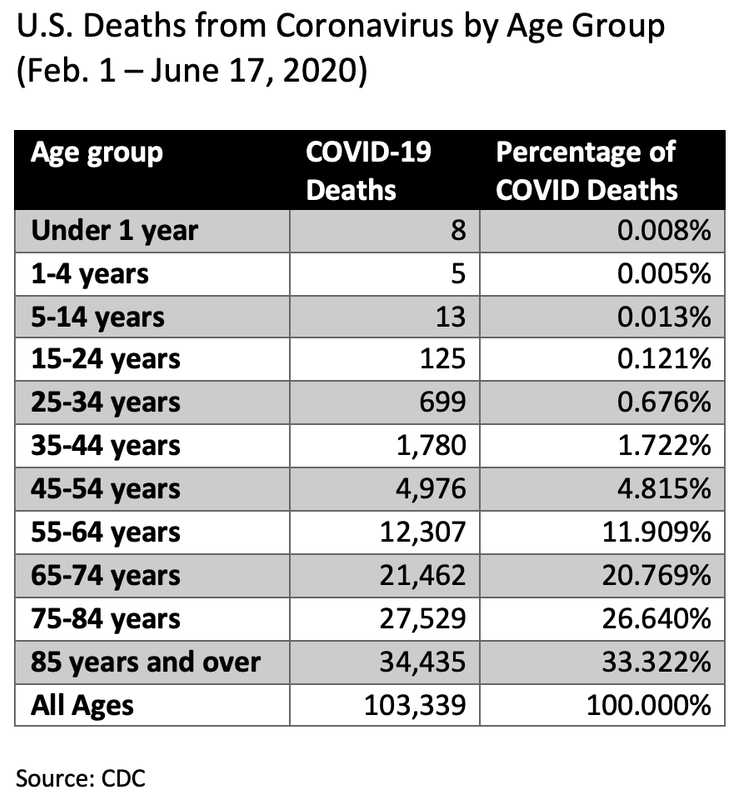
Why are we not getting a break down of confirmed deaths vs probable deaths?
Confirmed & Probable Counts
As of April 14, 2020, CDC case counts and death counts include both confirmed and probable cases and deaths. This change was made to reflect an
interim COVID-19 position statementpdf iconexternal icon issued by the Council for State and Territorial Epidemiologists on April 5, 2020. The position statement included a case definition and made COVID-19 a nationally notifiable disease. Nationally notifiable disease cases are voluntarily reported to CDC by jurisdictions.
A
confirmed case or death is defined by meeting confirmatory laboratory evidence for COVID-19.
A
probable case or death is defined by one of the following:
- Meeting clinical criteria AND epidemiologic evidence with no confirmatory laboratory testing performed for COVID-19
- Meeting presumptive laboratory evidence AND either clinical criteria OR epidemiologic evidence
- Meeting vital records criteria with no confirmatory laboratory testing performed for COVID19
Not all jurisdictions report probable cases and deaths to CDC. When not available to CDC, it is noted as N/A. Please note that jurisdictions may reclassify probable cases at any time to confirmed cases (if confirmatory laboratory evidence is obtained) or withdraw probable case reports entirely if further public health investigation determines that the individual most likely did not have COVID-19. As a result, probable case counts can fluctuate substantially. A jurisdiction might even report a negative number of probable cases on a given day, if more probable cases were disproven than were initially reported on that day.
You left out a bunch!! From your link...on criteria for covid....
Clinical Criteria
At least two of the following symptoms: fever (measured or subjective), chills, rigors, myalgia, headache, sore throat, new olfactory and taste disorder(s)
OR
At least one of the following symptoms: cough, shortness of breath, or difficulty breathing
OR
Severe respiratory illness with at least one of the following:
Clinical or radiographic evidence of pneumonia, OR
Acute respiratory distress syndrome (ARDS).
AND
No alternative more likely diagnosis
Laboratory Criteria
Laboratory evidence using a method approved or authorized by the U.S. Food and Drug Administration (FDA) or designated authority:
Confirmatory laboratory evidence:
Detection of severe acute respiratory syndrome coronavirus 2 ribonucleic acid (SARS-CoV-2 RNA) in a clinical specimen using a molecular amplification detection test
Presumptive laboratory evidence:
Detection of specific antigen in a clinical specimen
Detection of specific antibody in serum, plasma, or whole blood indicative of a new or recent infection*
*Serologic methods for diagnosis are currently being defined.
Epidemiologic Linkage
One or more of the following exposures in the 14 days before onset of symptoms:
Close contact** with a confirmed or probable case of COVID-19 disease; OR
Close contact** with a person with:
clinically compatible illness AND
linkage to a confirmed case of COVID-19 disease.
Travel to or residence in an area with sustained, ongoing community transmission of SARS-CoV-2.
Member of a risk cohort as defined by public health authorities during an outbreak.
**Close contact is defined as being within 6 feet for at least a period of 10 minutes to 30 minutes or more depending upon the exposure. In healthcare settings, this may be defined as exposures of greater than a few minutes or more. Data are insufficient to precisely define the duration of exposure that constitutes prolonged exposure and thus a close contact.
Criteria to Distinguish a New Case from an Existing Case
Not applicable (N/A) until more virologic data are available.
Case Classification
Probable
Meets clinical criteria AND epidemiologic evidence with no confirmatory laboratory testing performed for COVID-19.
Meets presumptive laboratory evidence AND either clinical criteria OR epidemiologic evidence.
Meets vital records criteria with no confirmatory laboratory testing performed for COVID-19.
Confirmed
Meets confirmatory laboratory evidence.
Other Criteria
Vital Records Criteria
A death certificate that lists COVID-19 disease or SARS-CoV-2 as a cause of death or a significant condition contributing to death.
Reference(s)
The Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. The Epidemiological Characteristics of an Outbreak of 2019 Novel Coronavirus Diseases (COVID-19) in China. Zhonghua Liu Xing Bing Xue Za Zhi. 2020;41(2):145–151. DOI:10.3760/cma.j.issn.0254-6450.2020.02.003.
Serial interval of novel coronavirus (COVID-19) infections. Hiroshi Nishiura, Natalie M. Linton, Andrei R. Akhmetzhanov PII: S1201-9712(20)30119-3 DOI:
Redirecting Reference: IJID 4006 To appear in: International Journal of Infectious Diseases, Accepted Date: 27 February 2020
Presymptomatic Transmission of SARS-CoV-2 — Singapore, January 23–March 16, 2020. Morbidity and Mortality Weekly Report Early Release / Vol. 69 April 1, 2020 U.S. Department of Health and Human Services Centers for Disease Control and Prevention. Wycliffe E. Wei; Zongbin Li; Calvin J. Chiew; Sarah E. Yong; Matthias P. Toh; Vernon J. Lee