How do we know more CO2 is causing warming?
.Must Go Up
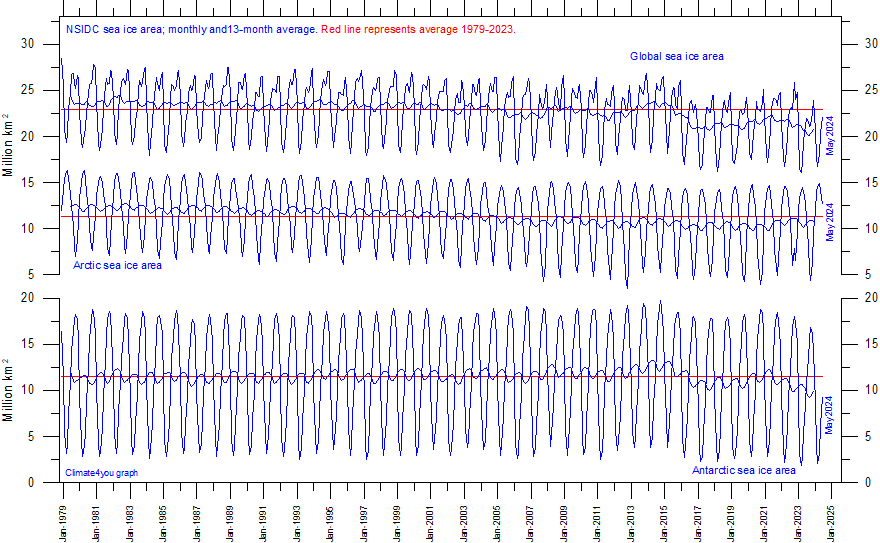
How long has CO2 been contributing to increased warming? According to NASA, Two-thirds of the warming has occurred since 1975. Is there a reliable way to identify CO2s influence on temperatures over that period?
There is: we can measure the wavelengths of long-wave radiation leaving the Earth (upward radiation). Satellites have recorded the Earth's outbound radiation. We can examine the spectrum of upward long-wave radiation in 1970 and 1997 to see if there are changes.
http://jvarekamp.web.wesleyan.edu/CO2/FP-1.pdf
Abstract
This experiment explored the absorptivity of four peaks, 1437, 1955, 2013, and
2060 nanometers, in the near-IR (NIR) absorption spectrum of CO2. The NIR absorption
bands in CO2 can contribute up to 30% of the total solar heating in the mesosphere.
Between the heights of 60-85 km the heating can exceed 1 K/day. (Fomichev & Shved,
1988; Ogibalov & Fomichev, 2003; Fomichev et al., 2004) With CO2 concentrations
increasing (West, 2005) it is ever more important to understand the absorbance properties
of this molecule in all of its absorbance bands. Modeling of the Beer Lambert law found
the absorption coefficient at 1955 nanometers to be 0.25 m2 mol-1 and the absorption
coefficient for the peaks at 2013 and 2060 nanometers to be 0.43 m2 mol-1. The
absorption peak at 1437 nanometers had an absorption coefficient >>10.
And still no datasets with source code to prove that CO2 drives climate other than AGW church propaganda.
And as already pointed out that CO2 does NOT drive climate is a reality despite the effort of the scribes of the AGW church to say otherwise.
<...snip...>
The main point is that every time you hear or read that CO2 heats the atmosphere , that energy is trapped by CO2 , that energy is stored by green house gases and similar statements , you may be sure that this source is not to be trusted for information about radiation questions .
------
The translation-translation process (sphere collision) has been well understood since more than 100 years . It can be studied by semi-classical statistical mechanics and the result is that the velocities of molecules (translational kinetic energy) within a volume of gas in equilibrium are distributed according to the Maxwell-Boltzmann distribution . As this distribution is invariant for a constant temperature , there are no net energy transfers and we do not need to further analyze this process .
The 2 processes of interest are the following :
CO2 + γ → CO2* (1)
This reads a CO2 molecule absorbs an infrared photon γ and goes to a vibrationally excited state CO2*
CO2* + N2 → CO2 + N2⁺ (2)
This reads a vibrationally excited CO2 molecule CO2* collides with an N2 molecule and relaxes to a lower vibrational energy state CO2 while the N2 molecule increases its velocity to N2⁺ . We use a different symbol * and ⁺ for the excited states to differentiate the energy modes vibrational (*) for CO2 and translational (⁺

for N2 . In other words , there is transfer between vibrational and translational degrees of freedom in the process (2) . This process in non equilibrium conditions is sometimes called thermalization .
The microscopical process (2) is described by time symmetrical equations . All mechanical and electromagnetical interactions are governed by equations invariant under time reversal . This is not true for electroweak interactions but they play no role in the process (2) .
Again in simple words , it means that if the process (2) happens then the time symmetrical process , namely CO2 + N2⁺ → CO2* + N2 , happens too . Indeed this time reversed process where fast (e.g hot) N2 molecules slow down and excite vibrationally CO2 molecules is what makes an N2/CO2 laser work. Therefore the right way to write the process (2) is the following .
CO2* + N2 ↔ CO2 + N2⁺ (3)
Where the use of the double arrow ↔ instad of the simple arrow → is telling us that this process goes in both directions . Now the most important question is What are the rates of the → and the ← processes ?
The LTE conditions with the energy equipartition law give immediately the answer : These rates are exactly equal . This means that for every collision where a vibrationally excited CO2* transfers energy to N2 , there is a collision where N2⁺ transfers the same energy to CO2 and excites it vibrationally . There is no net energy transfer from CO2 to N2 through the vibration-translation interaction .
As we have seen that CO2 cannot transfer energy to N2 through the translation-translation process either , there is no net energy transfer (e.g heating) from CO2 to N2 what proves our statement .
This has an interesting corollary for the process (1) , IR absorption by CO2 molecules . We know that in equilibrium the distribution of the vibrational quantum states (e.g how many molecules are in a state with energy Ei) is invariant and depends only on temperature . For example only about 5 % of CO2 molecules are in a vibrationally excited state at room temperatures , 95 % are in the ground state .
Therefore in order to maintain the number of vibrationally excited molecules constant , every time a CO2 molecule absorbs an infrared photon and excites vibrationally , it is necessary that another CO2 molecule relaxes by going to a lower energy state . As we have seen above that this relaxation cannot happen through collisions with N2 because no net energy transfer is permitted , only the process (1) is available . Indeed the right way to write the process (1) is also :
CO2 + γ ↔ CO2* (1)
Where the use of the double arrow shows that the absorption process (→

happens at the same time as the emission process (←

. Because the number of excited molecules in a small volume in LTE must stay constant , follows that both processes emission/absorption must balance . In other words CO2 which absorbs strongly the 15µ IR , will emit strongly almost exactly as much 15 µ radiation as it absorbs . This is independent of the CO2 concentrations and of the intensity of IR radiation .
For those who prefer experimental proofs to theoretical arguments , here is a simple experiment demonstrating the above statements . Let us consider a hollow sphere at 15°C filled with air . You install an IR detector on the surface of the cavity . This is equivalent to the atmosphere during the night . The cavity will emit IR according to a black body law . Some frequencies of this BB radiation will be absorbed by the vibration modes of the CO2 molecules present in the air . What you will observe is :
◾The detector shows that the cavity absorbs the same power on 15µ as it emits
◾The temperature of the air stays at 15°C and more specifically the N2 and O2 do not heat
These observations demonstrate as expected that CO2 emits the same power as it absorbs and that there is no net energy transfer between the vibrational modes of CO2 and the translational modes of N2 and O2 . If you double the CO2 concentration or make the temperature vary , the observations stay identical showing that the conclusions we made are independent of temperatures and CO2 concentrations .
CO2 heats the atmosphere?a counter view | Watts Up With That?