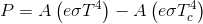Having to do with radiative heat transfer
Let's start with Wikipedia:
Radiation
Thermal radiation occurs through a vacuum or any transparentmedium (solid orfluid). It is the transfer of energy by means of photons inelectromagnetic wavesgoverned by the same laws.[14]Earth's radiation balance depends on the incoming and the outgoing thermal radiation, Earth's energy budget. Anthropogenicperturbations in the climate system, are responsible for a positive radiative forcingwhich reduces the net longwave radiation loss out to Space.
Thermal radiation is energy emitted by matter as electromagnetic waves, due to the pool of thermal energy in all matter with a temperature above absolute zero. Thermal radiation propagates without the presence of matter through the vacuum of space.[15]
Thermal radiation is a direct result of the random movements of atoms and molecules in matter. Since these atoms and molecules are composed of charged particles (protons and electrons), their movement results in the emission ofelectromagnetic radiation, which carries energy away from the surface.
The Stefan-Boltzmann equation, which describes the rate of transfer of radiant energy, is as follows for an object in a vacuum :
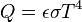
For radiative transfer between two objects, the equation is as follows:
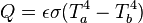
where Q is the rate of heat transfer, ε is the emissivity (unity for a black body), σ is the Stefan-Boltzmann constant, and T is the absolute temperature (in Kelvin or Rankine). Radiation is typically only important for very hot objects, or for objects with a large temperature difference.
Radiation from the sun, or solar radiation, can be harvested for heat and power.[16]Unlike conductive and convective forms of heat transfer, thermal radiation can be concentrated in a small spot by using reflecting mirrors, which is exploited inconcentrating solar power generation.[17] For example, the sunlight reflected from mirrors heats the PS10 solar power tower and during the day it can heat water to 285 °C (545 °F).
*************************************************************************************************
I know Todd had these equations up here before. I'm just curious how you explain the TWO TEMPERATURES in the equation for heat transfer between two objects. I would have thought that in your world, the difference between the two temperatures would simply act as some sort of gate. Like: If Ta>Tb then [Q=sigma deltaTa] else [Q=0]. Right? But that's NOT what they've got there. What they have there shows that the heat transfer actually taking place is the net, the algebraic sum, of the heat being transferred from hot to cold and from cold to hot.
I believe Todd pointed this one out to you as well. Let's say I have three cannonballs. One is 100C, the next is 50C. The last one is 0C. I have an insulated chamber in which I can place two of these at a time and monitor what their temperatures do. I first put the 100C in with the 50C. The first ball cools while the second ball warms up. I take them out and bring the 100C ball back up to temperature and then put it in with the 0C ball. Again, the hot ball cools and the cool ball warms.
What's the difference? Ta^4-Tb^4 is larger in the second case than in the first. So Q, the rate of exchange, the rate of heat transfer is much faster in the second case than in the first. The RATE at which heat is transferred is dependent on the magnitude of the temperature DIFFERENCE. The total transfer is the NET result of transfer taking place in BOTH DIRECTIONS. What you have been suggesting is that the hot ball will radiate as single-body Stefen-Boltzman says 100C will radiate and the second ball will not radiate at all. There would be no difference in the heat transfer rate in the two cases.
Do you disagree?
Let's start with Wikipedia:
Radiation
Thermal radiation occurs through a vacuum or any transparentmedium (solid orfluid). It is the transfer of energy by means of photons inelectromagnetic wavesgoverned by the same laws.[14]Earth's radiation balance depends on the incoming and the outgoing thermal radiation, Earth's energy budget. Anthropogenicperturbations in the climate system, are responsible for a positive radiative forcingwhich reduces the net longwave radiation loss out to Space.
Thermal radiation is energy emitted by matter as electromagnetic waves, due to the pool of thermal energy in all matter with a temperature above absolute zero. Thermal radiation propagates without the presence of matter through the vacuum of space.[15]
Thermal radiation is a direct result of the random movements of atoms and molecules in matter. Since these atoms and molecules are composed of charged particles (protons and electrons), their movement results in the emission ofelectromagnetic radiation, which carries energy away from the surface.
The Stefan-Boltzmann equation, which describes the rate of transfer of radiant energy, is as follows for an object in a vacuum :
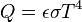
For radiative transfer between two objects, the equation is as follows:
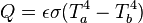
where Q is the rate of heat transfer, ε is the emissivity (unity for a black body), σ is the Stefan-Boltzmann constant, and T is the absolute temperature (in Kelvin or Rankine). Radiation is typically only important for very hot objects, or for objects with a large temperature difference.
Radiation from the sun, or solar radiation, can be harvested for heat and power.[16]Unlike conductive and convective forms of heat transfer, thermal radiation can be concentrated in a small spot by using reflecting mirrors, which is exploited inconcentrating solar power generation.[17] For example, the sunlight reflected from mirrors heats the PS10 solar power tower and during the day it can heat water to 285 °C (545 °F).
*************************************************************************************************
I know Todd had these equations up here before. I'm just curious how you explain the TWO TEMPERATURES in the equation for heat transfer between two objects. I would have thought that in your world, the difference between the two temperatures would simply act as some sort of gate. Like: If Ta>Tb then [Q=sigma deltaTa] else [Q=0]. Right? But that's NOT what they've got there. What they have there shows that the heat transfer actually taking place is the net, the algebraic sum, of the heat being transferred from hot to cold and from cold to hot.
I believe Todd pointed this one out to you as well. Let's say I have three cannonballs. One is 100C, the next is 50C. The last one is 0C. I have an insulated chamber in which I can place two of these at a time and monitor what their temperatures do. I first put the 100C in with the 50C. The first ball cools while the second ball warms up. I take them out and bring the 100C ball back up to temperature and then put it in with the 0C ball. Again, the hot ball cools and the cool ball warms.
What's the difference? Ta^4-Tb^4 is larger in the second case than in the first. So Q, the rate of exchange, the rate of heat transfer is much faster in the second case than in the first. The RATE at which heat is transferred is dependent on the magnitude of the temperature DIFFERENCE. The total transfer is the NET result of transfer taking place in BOTH DIRECTIONS. What you have been suggesting is that the hot ball will radiate as single-body Stefen-Boltzman says 100C will radiate and the second ball will not radiate at all. There would be no difference in the heat transfer rate in the two cases.
Do you disagree?