Unless energy is added back into the system, it absolutely will.Never happen!Energy flows from "hot" (high energy regions) to "cold" (lower energy regions).You violate the SLoT. In the real SLoT the change in entropy can equal zero, in your fake Creationist "SLoT" it can't.I explained it as clearly as I can. Can you tell me which part of my explanation was wrong and why?Double-talk does not substitute for math, you cannot eliminate the "equal to zero" part in the EQUATION with words, you MUST show the math.No. It absolutely has nothing to do with useable energy being lost during every matter to energy conversion. There are no matter to energy conversions that are 100% efficient.
So the universe starts out perfectly ordered with an entropy of zero. During each matter to energy conservation useable energy is lost because the process is not 100% efficient. At no time can this energy be regained as useable. If there were absolutely no processes going on then the change in entropy could be zero but the total entropy would still be greater than zero. But there are always conversions going on in the universe. So the change in entropy is always greater than zero.
Try again.

Entropy is the spreading out of energy.
Let's take two regions as an example. As energy becomes evenly distributed across two regions, the temperature of the two regions becomes equal.
The same thing is happening on a universal scale. The Sun and every other star are radiating energy into the universe.
However, they can’t do it forever. Eventually the stars will cool down, and heat will have spread out so much that there won’t be warmer objects and cooler objects.
Everything will be the same temperature.
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.

Note: This feature currently requires accessing the site using the built-in Safari browser.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Bull Ring ding: Does work in physics require motion
- Thread starter edthecynic
- Start date
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #222
Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!The universe can't help but to lose useable energy, Ed, unless something is adding energy to the system.No it didn't, in fact the matter at the outer edges of the universe is ACCELERATING away from us. If the universe was losing energy it would be slowing down!!!!!Once the universe began expanding it did begin losing useable energy, Ed. Wasn't that clear?If the universe has an entropy of zero then it never loses energy. The electron orbiting the nucleus of a stable atom has an entropy of zero, it never loses energy, if it did then it would be drawn into the nucleus, splitting it and no matter could exist.So the universe starts out perfectly ordered with an entropy of zero.
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #223
It absolutely won't.Unless energy is added back into the system, it absolutely will.Never happen!Energy flows from "hot" (high energy regions) to "cold" (lower energy regions).You violate the SLoT. In the real SLoT the change in entropy can equal zero, in your fake Creationist "SLoT" it can't.I explained it as clearly as I can. Can you tell me which part of my explanation was wrong and why?Double-talk does not substitute for math, you cannot eliminate the "equal to zero" part in the EQUATION with words, you MUST show the math.
Try again.

Entropy is the spreading out of energy.
Let's take two regions as an example. As energy becomes evenly distributed across two regions, the temperature of the two regions becomes equal.
The same thing is happening on a universal scale. The Sun and every other star are radiating energy into the universe.
However, they can’t do it forever. Eventually the stars will cool down, and heat will have spread out so much that there won’t be warmer objects and cooler objects.
Everything will be the same temperature.
I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!The universe can't help but to lose useable energy, Ed, unless something is adding energy to the system.No it didn't, in fact the matter at the outer edges of the universe is ACCELERATING away from us. If the universe was losing energy it would be slowing down!!!!!Once the universe began expanding it did begin losing useable energy, Ed. Wasn't that clear?If the universe has an entropy of zero then it never loses energy. The electron orbiting the nucleus of a stable atom has an entropy of zero, it never loses energy, if it did then it would be drawn into the nucleus, splitting it and no matter could exist.So the universe starts out perfectly ordered with an entropy of zero.
How exactly will that happen if there are no processes that are 100% efficient?It absolutely won't.Unless energy is added back into the system, it absolutely will.Never happen!Energy flows from "hot" (high energy regions) to "cold" (lower energy regions).You violate the SLoT. In the real SLoT the change in entropy can equal zero, in your fake Creationist "SLoT" it can't.I explained it as clearly as I can. Can you tell me which part of my explanation was wrong and why?
Entropy is the spreading out of energy.
Let's take two regions as an example. As energy becomes evenly distributed across two regions, the temperature of the two regions becomes equal.
The same thing is happening on a universal scale. The Sun and every other star are radiating energy into the universe.
However, they can’t do it forever. Eventually the stars will cool down, and heat will have spread out so much that there won’t be warmer objects and cooler objects.
Everything will be the same temperature.
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #226
It contradicts your fake SLoT.I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!The universe can't help but to lose useable energy, Ed, unless something is adding energy to the system.No it didn't, in fact the matter at the outer edges of the universe is ACCELERATING away from us. If the universe was losing energy it would be slowing down!!!!!Once the universe began expanding it did begin losing useable energy, Ed. Wasn't that clear?If the universe has an entropy of zero then it never loses energy. The electron orbiting the nucleus of a stable atom has an entropy of zero, it never loses energy, if it did then it would be drawn into the nucleus, splitting it and no matter could exist.
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #227
That's a lie, I already gave you an example of the change in entropy equaling zero.How exactly will that happen if there are no processes that are 100% efficient?It absolutely won't.Unless energy is added back into the system, it absolutely will.Never happen!Energy flows from "hot" (high energy regions) to "cold" (lower energy regions).You violate the SLoT. In the real SLoT the change in entropy can equal zero, in your fake Creationist "SLoT" it can't.
Entropy is the spreading out of energy.
Let's take two regions as an example. As energy becomes evenly distributed across two regions, the temperature of the two regions becomes equal.
The same thing is happening on a universal scale. The Sun and every other star are radiating energy into the universe.
However, they can’t do it forever. Eventually the stars will cool down, and heat will have spread out so much that there won’t be warmer objects and cooler objects.
Everything will be the same temperature.
Can you explain how it does that?It contradicts your fake SLoT.I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!The universe can't help but to lose useable energy, Ed, unless something is adding energy to the system.No it didn't, in fact the matter at the outer edges of the universe is ACCELERATING away from us. If the universe was losing energy it would be slowing down!!!!!Once the universe began expanding it did begin losing useable energy, Ed. Wasn't that clear?
Are you saying that applies to everything? That every matter to energy exchange is 100% efficient?That's a lie, I already gave you an example of the change in entropy equaling zero.How exactly will that happen if there are no processes that are 100% efficient?It absolutely won't.Unless energy is added back into the system, it absolutely will.Never happen!Energy flows from "hot" (high energy regions) to "cold" (lower energy regions).
Entropy is the spreading out of energy.
Let's take two regions as an example. As energy becomes evenly distributed across two regions, the temperature of the two regions becomes equal.
The same thing is happening on a universal scale. The Sun and every other star are radiating energy into the universe.
However, they can’t do it forever. Eventually the stars will cool down, and heat will have spread out so much that there won’t be warmer objects and cooler objects.
Everything will be the same temperature.
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #230
Your fake SLoT says everything in the universe is losing energy and therefore slowing down, but we can measure the expansion of the universe and matter close to us IS slowing down, but at the extreme edges of the universe matter is speeding up and therefore gaining energy.Can you explain how it does that?It contradicts your fake SLoT.I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!The universe can't help but to lose useable energy, Ed, unless something is adding energy to the system.No it didn't, in fact the matter at the outer edges of the universe is ACCELERATING away from us. If the universe was losing energy it would be slowing down!!!!!
Of course, as you know the universe is NOT a closed system. If there is no multiverse, then it is an isolated system, but as far as the visible universe goes it is an open system with or without a multiverse.
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #231
Geeezzzz, how many times do you have to be shown the equation for the actual real SLoT before it sinks in??????Are you saying that applies to everything? That every matter to energy exchange is 100% efficient?That's a lie, I already gave you an example of the change in entropy equaling zero.How exactly will that happen if there are no processes that are 100% efficient?It absolutely won't.Unless energy is added back into the system, it absolutely will.Never happen!

Everything I have written assumes the universe is an isolated system. And by you assuming the "visible" universe is an open system and that the universe is gaining energy, then something must be putting energy into the system, right?Your fake SLoT says everything in the universe is losing energy and therefore slowing down, but we can measure the expansion of the universe and matter close to us IS slowing down, but at the extreme edges of the universe matter is speeding up and therefore gaining energy.Can you explain how it does that?It contradicts your fake SLoT.I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!The universe can't help but to lose useable energy, Ed, unless something is adding energy to the system.
Of course, as you know the universe is NOT a closed system. If there is no multiverse, then it is an isolated system, but as far as the visible universe goes it is an open system with or without a multiverse.
I have already explained this equation to you almost a dozen times. It doesn't say what you are saying. It says you can never reduce entropy. There are no processes that are 100% efficient. The change in entropy for the universe cannot be zero until the universe has reached maximum entropy. Energy flows from "hot" (high energy regions) to "cold" (lower energy regions). Entropy is the spreading out of energy. Unless energy is added to the system - which seems to be what you are saying - eventually the stars will cool down, and heat will have spread out so much that there won’t be warmer objects and cooler objects. Everything will be the same temperature.Geeezzzz, how many times do you have to be shown the equation for the actual real SLoT before it sinks in??????Are you saying that applies to everything? That every matter to energy exchange is 100% efficient?That's a lie, I already gave you an example of the change in entropy equaling zero.How exactly will that happen if there are no processes that are 100% efficient?It absolutely won't.Unless energy is added back into the system, it absolutely will.

Entropy and the second law
The second law of thermodynamics is one of the most fundamental laws of nature, having profound implications. In essence, it says this:
The second law - The level of disorder in the universe is steadily increasing. Systems tend to move from ordered behavior to more random behavior.
One implication of the second law is that heat flows spontaneously from a hotter region to a cooler region, but will not flow spontaneously the other way. This applies to anything that flows: it will naturally flow downhill rather than uphill.
If you watched a film forwards and backwards, you would almost certainly be able to tell which way was which because of the way things happen. A pendulum will gradually lose energy and come to a stop, but it doesn't pick up energy spontaneously; an ice cube melts to form a puddle, but a puddle never spontaneously transforms itself into an ice cube; a glass falling off a table might shatter when it hits the ground, but the pieces will never spontaneously jump back together to form the glass again. Many processes are irreversible, and any irreversible process increases the level of disorder. One of the most important implications of the second law is that it indicates which way time goes - time naturally flows in a way that increases disorder.
The second law also predicts the end of the universe: it implies that the universe will end in a "heat death" in which everything is at the same temperature. This is the ultimate level of disorder; if everything is at the same temperature, no work can be done, and all the energy will end up as the random motion of atoms and molecules.
The second law of thermodynamics is one of the most fundamental laws of nature, having profound implications. In essence, it says this:
The second law - The level of disorder in the universe is steadily increasing. Systems tend to move from ordered behavior to more random behavior.
One implication of the second law is that heat flows spontaneously from a hotter region to a cooler region, but will not flow spontaneously the other way. This applies to anything that flows: it will naturally flow downhill rather than uphill.
If you watched a film forwards and backwards, you would almost certainly be able to tell which way was which because of the way things happen. A pendulum will gradually lose energy and come to a stop, but it doesn't pick up energy spontaneously; an ice cube melts to form a puddle, but a puddle never spontaneously transforms itself into an ice cube; a glass falling off a table might shatter when it hits the ground, but the pieces will never spontaneously jump back together to form the glass again. Many processes are irreversible, and any irreversible process increases the level of disorder. One of the most important implications of the second law is that it indicates which way time goes - time naturally flows in a way that increases disorder.
The second law also predicts the end of the universe: it implies that the universe will end in a "heat death" in which everything is at the same temperature. This is the ultimate level of disorder; if everything is at the same temperature, no work can be done, and all the energy will end up as the random motion of atoms and molecules.
Entropy and the second law
A measure of the level of disorder of a system is entropy, represented by S. Although it's difficult to measure the total entropy of a system, it's generally fairly easy to measure changes in entropy. For a thermodynamic system involved in a heat transfer of size Q at a temperature T , a change in entropy can be measured by:
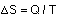
The second law of thermodynamics can be stated in terms of entropy. If a reversible process occurs, there is no net change in entropy. In an irreversible process, entropy always increases, so the change in entropy is positive. The total entropy of the universe is continually increasing.
A measure of the level of disorder of a system is entropy, represented by S. Although it's difficult to measure the total entropy of a system, it's generally fairly easy to measure changes in entropy. For a thermodynamic system involved in a heat transfer of size Q at a temperature T , a change in entropy can be measured by:
The second law of thermodynamics can be stated in terms of entropy. If a reversible process occurs, there is no net change in entropy. In an irreversible process, entropy always increases, so the change in entropy is positive. The total entropy of the universe is continually increasing.
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #237
The SLoT is for CLOSED systems.Everything I have written assumes the universe is an isolated system. And by you assuming the "visible" universe is an open system and that the universe is gaining energy, then something must be putting energy into the system, right?Your fake SLoT says everything in the universe is losing energy and therefore slowing down, but we can measure the expansion of the universe and matter close to us IS slowing down, but at the extreme edges of the universe matter is speeding up and therefore gaining energy.Can you explain how it does that?It contradicts your fake SLoT.I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?Well something is accelerating matter at the outer edges of the universe, or do you deny even that!!!
Of course, as you know the universe is NOT a closed system. If there is no multiverse, then it is an isolated system, but as far as the visible universe goes it is an open system with or without a multiverse.
How else can you explain matter at the extreme edges of the visible universe accelerating????
And it is not an assumption, it is a measured fact!!!!
edthecynic
Censored for Cynicism
- Oct 20, 2008
- 43,044
- 6,883
- 1,830
- Thread starter
- #238
Entropy and the second law
A measure of the level of disorder of a system is entropy, represented by S. Although it's difficult to measure the total entropy of a system, it's generally fairly easy to measure changes in entropy. For a thermodynamic system involved in a heat transfer of size Q at a temperature T , a change in entropy can be measured by:
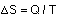
The second law of thermodynamics can be stated in terms of entropy. If a reversible process occurs, there is no net change in entropy. In an irreversible process, entropy always increases, so the change in entropy is positive. The total entropy of the universe is continually increasing.
Entropy and the 2nd Law in Open SystemsThe SLoT is for CLOSED systems.Everything I have written assumes the universe is an isolated system. And by you assuming the "visible" universe is an open system and that the universe is gaining energy, then something must be putting energy into the system, right?Your fake SLoT says everything in the universe is losing energy and therefore slowing down, but we can measure the expansion of the universe and matter close to us IS slowing down, but at the extreme edges of the universe matter is speeding up and therefore gaining energy.Can you explain how it does that?It contradicts your fake SLoT.I deny it has anything to do with the 2nd Law of Thermodynamics. Can you explain to me how it does?
Of course, as you know the universe is NOT a closed system. If there is no multiverse, then it is an isolated system, but as far as the visible universe goes it is an open system with or without a multiverse.
How else can you explain matter at the extreme edges of the visible universe accelerating????
And it is not an assumption, it is a measured fact!!!!
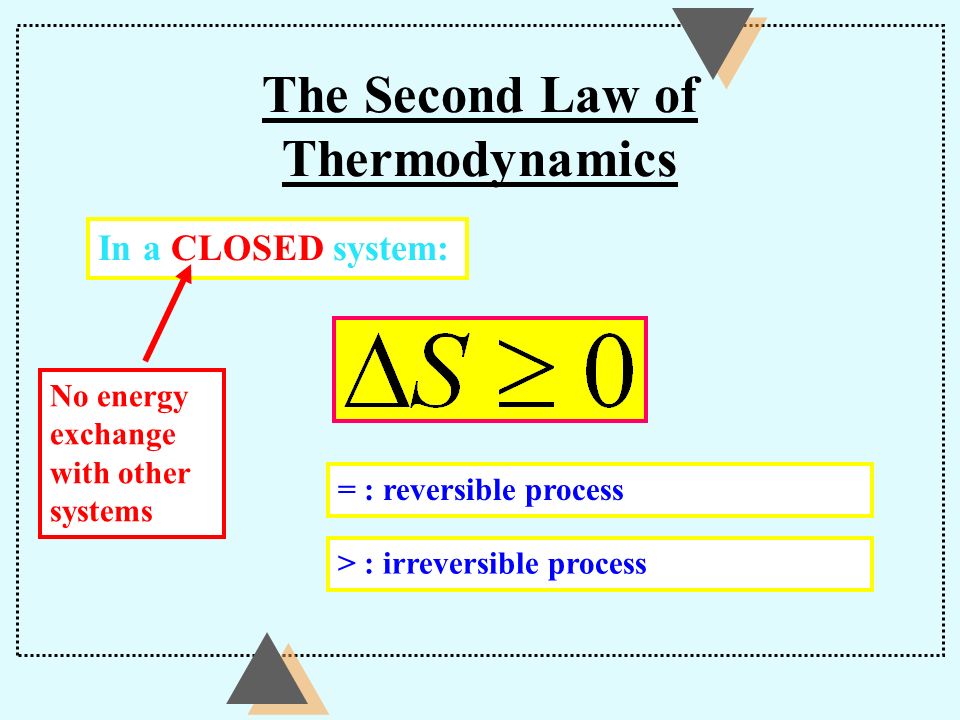
If anything can pass into, or out of, a system, we say it is an open system. If only matter can pass into, or out of, a system, but not energy, then we call it a closed system. If neither matter nor energy can pass into, or out of, a system, then we call it an isolated system.
We have a definition of the 2nd law from our previous chapter, a standard definition from standard thermodynamics.
Processes in which the entropy of an isolated system would decrease do not occur, or, in every process taking place in an isolated system, the entropy of the system either increases or remains constant. The definition explicitly requires the system in question to be isolated. This is a non trivial observation. If the system were not isolated, then entropy could pour out over the boundary, and the entropy decrease instead of increase.
So my question still stands. Are you assuming that energy is being added to the universe? Or is it that you are assuming the universe is an open system? I don't know anyone who assumes that.
Are you going to address the position from the physics department of Boston University?
And isolated systems too.The SLoT is for CLOSED systems.
Similar threads
- Replies
- 4
- Views
- 195
- Replies
- 2
- Views
- 143
- Replies
- 79
- Views
- 958
- Replies
- 8
- Views
- 164
Latest Discussions
- Replies
- 111
- Views
- 597
- Replies
- 147
- Views
- 624
Forum List
-
-
-
-
-
Political Satire 8058
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
ObamaCare 781
-
-
-
-
-
-
-
-
-
-
-
Member Usernotes 469
-
-
-
-
-
-
-
-
-
-