[/B]
If You knew anything about calculus then you`ld also know that there is no way that the temperature can be a
linear proportional function of the CO2 IR absorption which is a
non linear log function.
If You click on that:
Modtran Infrared Atmospheric Radiation Code
then run the model with CO2 starting @ 250 ppm then repeat to ~350 ppm or more and check the numeric output on the right side then You can see that the delta T increase the best ISDN computer model puts out
is a LINEAR 0.1 per 10 ppm CO2 increase....
which is exactly the point "flacaltenn" was making.!!!
There's no delta T on the right side. Sorry, please explain.
That`s a reasonable request,...Sure I`ll explain,because the delta T is not directly displayed by the Modtran. You have to obtain it Yourself by submitting ~ 10 different CO2 Modtran requests and then plot them as T versus ppm CO2.
Go to the Modtran, enter a value for CO2, click on the "submit calculation button", then on the right side Model Output window,
You have to scroll all the way down to see it, there is a button "View the whole output file"
click on that and then You got the numeric data which is used to plot the graphs You have just seen in this window.
But to get the delta T values You have to do that Yourself and use the Modtran in 10 ppm increments. Then You can use Your favorite "spread sheet", enter in the cell where You want to see dT=T2-T1, copy it down that column and enter the Modtran numeric data output...You do know how to use a spreadsheet right?
Even though it`s still tedious to do all that....funny that they don`t offer a Modtran version that does that and just give You the numeric output..the only thing they are really generous with are graphs with a totally disproportional Y-axis factor to really make the point how T skyrockets with ppm CO2 on the X-axis.
Anyway I did that 2 years ago, then again last year and the Temp. increase was a
constant step for each
10 ppm increment from 250 ppm all the way up to 500 ppm CO2...!!!
Maybe they finally "corrected" that,....these "re-adjustments" they do,...lately almost every week.
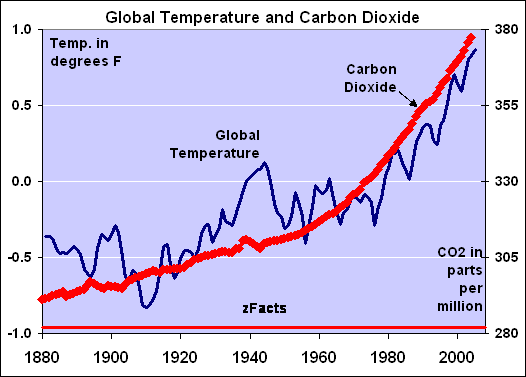
I got better things to do than running numbers through their stupid Modtran every day..You can save Yourself all that work and just look at A
NY "global warming" degT versus ppm CO2 GRAPH they published...
On every graph they published the
T-curve runs parallel with the ppm CO2,...the
temperature is DIRECTLY PROPORTIONAL to ppm CO2...and
All proportional functions are LINEAR...!!!
Don`t tell me You did not know that.
By the way, if You do want to go through all that trouble then while You are at it take a look how wildly the Temps vary when You enter different atmospheric conditions. They don`t even bother themselves to do that for the data they publish....not even at the point where data is collected.
..ppm CO2 are "adjusted" for standard atm conditions right at Mauna Loa and
every sampling point they have....Moisture, bar-pressure never even enters the equation already at that point...:
http://www.esrl.noaa.gov/gmd/ccgg/trends/
Monthly mean atmospheric carbon dioxide at Mauna Loa Observatory, Hawaii
The carbon dioxide data (red curve), measured as the mole fraction in dry air, on Mauna Loa constitute the longest record of direct measurements of CO2 in the atmosphere.
Data are reported as a dry mole fraction defined as the number of molecules of carbon dioxide divided by the number of molecules of dry air multiplied by one million (ppm).
And that`s the CO2 data used in EVERY COMPUTER MODEL...!!!
Of course, because if You use MOLAR ppm then the CO2 remains constant regardless of pressure...but if You do run an IR spectrophotometer that obeys Beer&Lambert`s absorption laws then it`s the absolute amount of CO2 per cubic meter air that matters...and that gets less and less with altitude.
The real cheat however is the "divided by the #of Mol.s DRY AIR"...not only because that already inflated the CO2 ppm by taking moisture out of the equation, but mostly because You can`t even claim while keeping a straight face that You could even measure an increase of 100 ppm CO2 in air at normal moisture with an infrared spectrophotometer!!! To do that You have to use GC (Gas Chromathography)....Like we did where I was:
You can see it front left, that`s a GC...and on that pic You can see another one on the left, smack center on the lab bench:
Inside is a column. You inject a precise amount of the sample gas and the gas components come out at different "rF" values. First the "rare" gasses in our atm, then Nitrogen & Oxygen, then the CO2 and after that whatever H2O was in the sample as "moisture". For a detector we used a "DTC" (differential thermal conductivity) and the whole thing is DEAD ACCURATE to a fraction of a ppm CO2....
which is why the Diesel Power Plant we run nearby to keep us alive made a complete joke out of the entire exercise..!!
It was part of my job to calibrate these instruments for that climate clown who was actually running the samples...he studied some Social Science crap...and that`s typical! When I came to Canada I worked first in the FDA, doing trace analysis,...meaning a shitload of IR and Atomic Absorption spectral analysis..then in the main RCMP forensic Lab when it was still in Winnipeg...got bored and joined the Military Engineers and landed in the Arctic.
In all these years I have never ever seen a real Chemist in any of these labs up in the arctic, and I bet at Mauna Loa it ain`t any different.