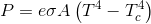I did...found this quote which settled the issue.
Second Law of Thermodynamics: It is not possible for
heat to flow from a colder body to a warmer body without any
work having been done to accomplish this flow. Energy will not flow spontaneously from a low
temperature object to a higher temperature object.
Now remind us what a true blue goober you are by claiming that the above statement only applies to refrigerators....or add claims that aren't stated in the law by whining that it is talking about net energy flows when it says nothing whatsoever about net energy flows..go ahead.
There are many ways of stating the second law of thermodynamics. Which ones of the following statements from universities or textbooks do you disagree with?
http://physics.about.com/od/thermodynamics/a/lawthermo_4.htm
It is impossible for a process to have as its sole result the transfer of heat from a cooler body to a hotter one.
LAWS OF THERMODYNAMICS
The Second Law of Thermodynamics states that "in all energy exchanges, if no energy enters or leaves the system, the potential energy of the state will always be less than that of the initial state."
Heat engines and the second law
The second law states that heat flows naturally from regions of higher temperature to regions of lower temperature, but that it will not flow naturally the other way.
Refrigerators
The Second Law of Thermodynamics states that heat will spontaneously always flow from a hot region to a cold region. By itself it never flows the other way, but can be made to do so under the influence of an external agency.
2nd Law of Thermodynamics
Heat flows spontaneously from a hot body ot a cool one.
http://physics.about.com/od/thermodynamics/a/lawthermo_4.htm
It is impossible for a process to have as its sole result the transfer of heat from a cooler body to a hotter one.
2nd Law of Thermodynamics
Nov 5, 2016 - The Second Law of Thermodynamics states that the state of entropy of the entire universe, as an isolated system, will always increase over time. The second law also states that the changes in the entropy in the universe can never be negative.
Blufton University
The Second Law of Thermodynamics
Heat will not flow spontaneously from a cold body to a hot body.
chemwiki.ucdavis.edu › ... › Laws of Thermodynamics
The Second Law of Thermodynamics states that the state of entropy of the entire universe, as an isolated system, will always increase over time. The second law also states that the changes in the entropy in the universe can never be negative.
MIT:
5.1 Concept and Statements of the Second Law
No process is possible whose sole result is the transfer of heat from a cooler to a hotter body.
Second Law: There exists for every thermodynamic system in equilibrium an extensive scalar property called the entropy, such that in an infinitesimal reversible change of state of the system, where is the absolute temperature and is the amount of heat received by the system. The entropy of a thermally insulated system cannot decrease and is constant if and only if all processes are reversible.
Book: "Thermodynamics." Cambridge university press.
There are many ways to introduce the Second Law of Thermodynamics...all of it's statements are equivalent and self-consistent....
"Heat flows spontaneously from a system of higher temperature to a system of lower temperature."....
Book: A course in Thermodynamics. Joseph Kestin
It is impossible to produce work by reducing the energy of a system existing in thermal equilibrium and confined within a rigid adiabatic enclosure.
second law of thermodynamics
Second law of thermodynamics - a law stating that mechanical work can be derived from a body only when that body interacts with another at a lower temperature; any spontaneous process results in an increase of entropy