Billy_Bob
Diamond Member
I asked you if you knew. It is basic atomic structure of atoms. I got taught this in high school AP chemistry.What is the electrical charge of a photon particle?Don't get all "sciency" on him...he is busy demonstrating that his comfort level doesn't extend beyond swapping insults...he is showing us that he can't interpret, in plain english, what even the most simple mathematical expression is saying.
Don't get all "sciency" on him...
"Covailent [sic] bonds repel photons" is sciency? DERP!
What is the electrical charge of a molecules covailent band? What is the frequency of that band? How will that frequency/charge affect a particle of opposite charge? of like charge?
There is a provable/empirically observed reason water will hold a photon and CO2 will not. There is a reason water will become excited and heat up, while CO2 will not.
What is the electrical charge of a photon particle?
Tell me.
What is the electrical charge of a molecules covailent band?
Tell me.
What is the frequency of that band? How will that frequency/charge affect a particle of opposite charge? of like charge?
Tell me.
There is a provable/empirically observed reason water will hold a photon and CO2 will not.
Hold a photon? With what?
There is a reason water will become excited and heat up, while CO2 will not.
What is that reason?
LOL... Basic Atomic Sciences taught to first year chemistry students.. DO some research.
You want me to research your idiotic claims? Nice try Sparky.
When you get a real source that says "covailent [sic] bonds repel photons from cooler matter", then I'll take your silliness more seriously.
Until then, your idiocy is damaging the anti-AGW argument. Moron.
How can this point damage a failed hypothesis? It clearly demonstrates that the CO2 monster is but a fallacy.
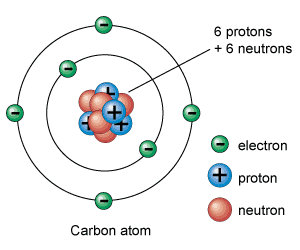
This doesn't show the magnetic bonds, but you can determine the covalent field strength of each layer and dominating polarity with what is shown.
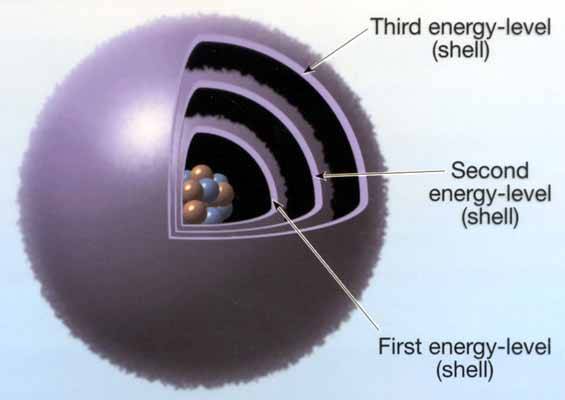
While simplistic, this gives you the basic picture of the varying fields.
Now tell me how a Photon, with a positive charge, will react to a low energy third level shell with a positive imbalance? (one or two negative charges vs 6 positive in the core) as is the case with CO. Remember like charges repel and if the predominate charge is positive what happens to the positively charged particle?
It is deflected..

Again the outer shell is positive and why photons are not held.
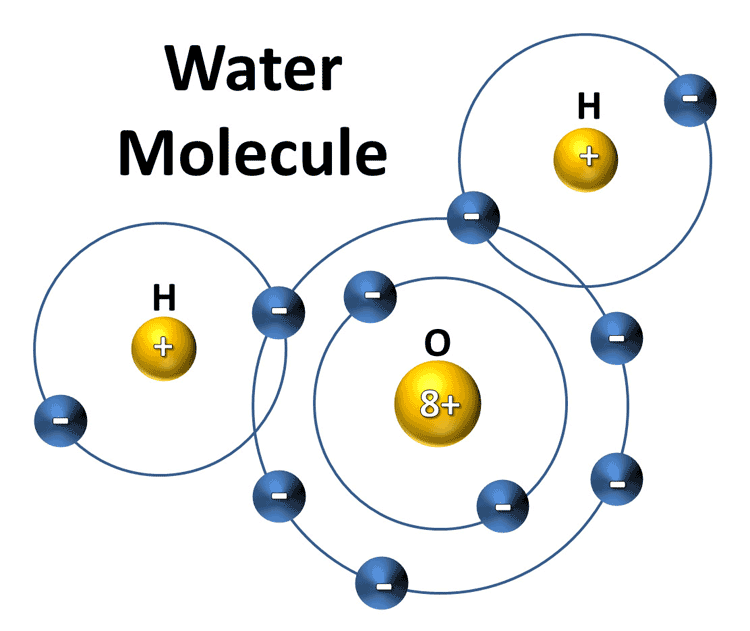
Water vapor's outer shell is NEGATIVE and the positive photon affects it by being held by it until it is dislodged by another photon or the molecule changes up due to chemical reaction (such as re-nucleating into a water droplet where the bonds share the outer bands making its cumulative charge positive repelling the photon particle).
Each of these molecules are resonating at their temperature values. This is important because that wavelength coupled with resonate frequency of the magnetic band can stop penetration of the outer shell. This is why there are bands of emission and absorption for each molecule.
Its pretty basic stuff..
The wave length of the dislodged photon is determined by the temperature of the molecule it was dislodged from.
A molecule resonating at a higher frequency will deflect a lower frequency photon. A warmer object will deflect a cooler objects radiated energy. Again this is shown in the absorption and emission bands of all matter.
Last edited: