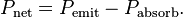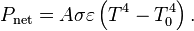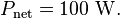WireBender:
Of course it's gonna re-radiate some of it. Maybe MOST of it at IR wavelengths. That's the "TRANSMISSION Quotient" shown in the graph. The question isn't how much filtering protection the CO2 offers from the sun's rays, but it's ability to deflect EM (IR) radiation FROM the surface -- back TO the surface or directed downwards in the atmosphere. OR to retain heat in the molecules themselves from either incident sunlight or surface reflected IR.
In the case of ::
Of course it "retains" it. Dry Ice warms and goes to it's vapor state. It only stays in gaseous state because it's RETAINING heat. At high altitude, the loss is pretty quick but that's thermodynamics driven. Just like the thermal diff that causes the stage effect for the witches.
Now here's the deal.. The heat can come from IR radiation OR it can come from any of the THERMAL transfer modes associated with heat transfer in materials. Even gases exchange heat thru conduction and convection for example (not just solids and liquids). I have many ways to make that Dry Ice smoke (by WARMING it)..
The question really goes back to your 1000W heater analogy. I can't CREATE additional heat from that heater, but could I INSULATE it with a gas that would marginally raise (say) the surface temp of that heater? I KNOW that's possible with a blanket because that's why the lawyers warn you in the instruction manual not to block the convection path of the heater. So given the fact that gases DO retain conductive/convective heat (not EM IR) -- I really don't see why you're so insistent on NO heat retention by the CO2. Any compound that can't "retain" heat is foreign to this universe..
BTW: those transmission curves I posted are not labeled for the transmission factors. But (100% - transmission factor) will give you an idea of heat energy retained.
Of course it's gonna re-radiate some of it. Maybe MOST of it at IR wavelengths. That's the "TRANSMISSION Quotient" shown in the graph. The question isn't how much filtering protection the CO2 offers from the sun's rays, but it's ability to deflect EM (IR) radiation FROM the surface -- back TO the surface or directed downwards in the atmosphere. OR to retain heat in the molecules themselves from either incident sunlight or surface reflected IR.
In the case of ::
Quote: Originally Posted by flacaltenn
I find it bizarre that you can't impart heat to CO2 ("or ANY OTHER GAS, other than water vapor"). It makes that opening scene in MacBeth with all the witches pure magic... How does it get from frozen witches brew to spewing around the stage?
WireBender:
I never said that you couldn't impart heat to the substance. I said that it wouldn't retain it.
Of course it "retains" it. Dry Ice warms and goes to it's vapor state. It only stays in gaseous state because it's RETAINING heat. At high altitude, the loss is pretty quick but that's thermodynamics driven. Just like the thermal diff that causes the stage effect for the witches.
Now here's the deal.. The heat can come from IR radiation OR it can come from any of the THERMAL transfer modes associated with heat transfer in materials. Even gases exchange heat thru conduction and convection for example (not just solids and liquids). I have many ways to make that Dry Ice smoke (by WARMING it)..
The question really goes back to your 1000W heater analogy. I can't CREATE additional heat from that heater, but could I INSULATE it with a gas that would marginally raise (say) the surface temp of that heater? I KNOW that's possible with a blanket because that's why the lawyers warn you in the instruction manual not to block the convection path of the heater. So given the fact that gases DO retain conductive/convective heat (not EM IR) -- I really don't see why you're so insistent on NO heat retention by the CO2. Any compound that can't "retain" heat is foreign to this universe..
BTW: those transmission curves I posted are not labeled for the transmission factors. But (100% - transmission factor) will give you an idea of heat energy retained.
Last edited: