You really don't know what he is talking about? Do you think a column of air has an equal pressure from the top to the bottom or do you think that the pressure at the bottom of the column has no effect on temperature and that the ideal gas laws are just so much bunk?
Here's a fine example of your perfect misapprehension of fundamental science: you and the ideal gas laws. You think a column of air will have a temperature gradient due to its pressure gradient. What a fine and discerning grasp of 8th grade science you possess. Unfortunately, in the 9th grade, apparently after you chose to drop out, they presented the idea of HEAT TRANSFER.
Think about how long has that column of air - the one in the Earth's atmosphere - been sitting there? Do you really believe it still holds a temperature gradient from its static pressure?
OR
Let me guess: you believe that pressure spontaneously and continuously
generates heat energy and that the higher pressure at the base of that column will generate more heat energy than will the lower pressure at its apex and that generation continues apace since the planet formed to this very day. Is that it?
As with almost every science topic you choose to address, your beliefs are so far from reality and so twisted by insane and incomprehensible nonsense, one knows not where to begin when attempting to correct you. Or what we might possibly owe you to motivate such beneficence.
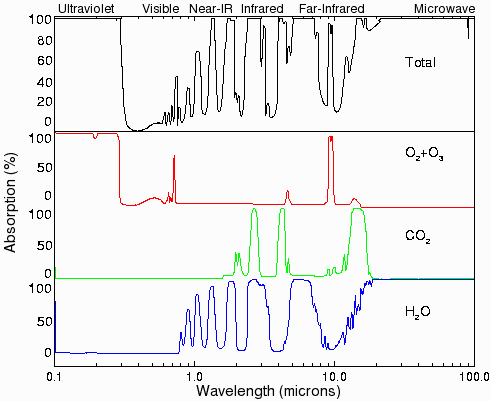
})
})