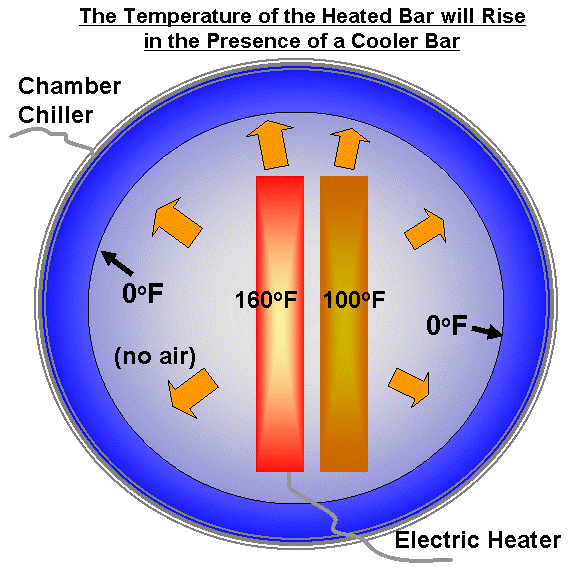
And nowhere do they say that the number of watts/m^2 AGW CO2 "back radiation" at 15µm depends on the air temperature.
Really? Now, I'm assuming that when you say "air" you do mean the air that contains the CO2 as part of its mix. But, then, you'd be saying that 800C air radiates no more energy than 8C air and I'm truly sorry to say this, but that's just wrong.
When you said that the number of watts emitted in the 15 um band was independent of temperature.
What`s the matter?...your little bird brain can`t figure out the difference between the black body radiation of a 800C hot gas which is all over the spectrum and what CO2 can absorb and re-emit as "back radiation" at very specific wave bands.
Next you should also find an AGW course for dummies, like "skepticalscience.com" or something like that and get them to explain it to you why the rest of the black body spectrum your 800 C hot blow torch air emits doesn`t matter, That`s because all the rest of the heat it radiates passes by the so called "atmospheric window" which is at 15µm....the one that is blocked by 400 ppm CO2.
1) The amount of radiation coming out of a volume of gas is directly dependent on its temperature (Planck's Law). As temperature rises, the energy under the spectrum curve,
including CO2's 15um absorption band, increases.
2) That CO2 absorbs and reemits in a narrow band does not
block the movement of radiation, it slows or smears it as radiation is absorbed and reemitted. Fifteen micron radiation does eventually reach the Earth's surface or escapes to space.
**** this is incredible how stupid you are.
You still don`t know the difference between C=O resonance which
absorbs and re-emits IR only at specific wavelength:
http://www.elmhurst.edu/%7Echm/vchembook/images/irCO2.JPEG
And black body radiation is
spread out over the entire spectrum like the one you copied and pasted which starts out at
3000 K and tops out at
5000K....also called a
continuous spectrum as the top one, as opposed to an absorption and emission spectrum below it:
http://astro.unl.edu/naap/hr/graphics/Spectral_lines_en.png
These absorption and emission lines are
specific to the substance at hand and have a
specific wavelength and intensity...that`s how spectroscopic analysis works....
and they don`t get more intense with increased temperature.
First you went to an 800 C extreme and now you are at 5000K to tell me that a black body radiates at any wavelength...as if I did not know or said otherwise and now you also added that
That CO2 absorbs and reemits in a narrow band does not block the movement of radiation, it slows or smears it as radiation is absorbed and reemitted.
Oh really, CO2
slows and "
smears" radiation ?
I`ve done a lot of IR spectral analysis and never noticed that.
You have to write a paper on that, because the rest of us are under the impression that it does block light from going straight through and re-emits whatever it has absorbed over a 360 deg steradian.
So can your little bird brain connect all these dots and figure out what would happen if you go to an 800 C blow torch temperature extreme?
...and that what your 800 C hot blow torch emits as IR has **** all to do any more with 15 µm IR absorption and re-emission.
Maybe you should watch another demonstration for dummies like you.
That candle isn`t 800 C hot but it`s hot enough:
[ame="http://www.youtube.com/watch?v=SeYfl45X1wo"]CO2 experiment - YouTube[/ame]
So where did all that IR from the hot candle go ?
According to you there should have been plenty more IR from the hot flame to make up for what the CO2 absorbed.
2) That CO2 absorbs and reemits in a narrow band does not block the movement of radiation, it slows or smears it as radiation is absorbed
Oh now I get it, what you are saying...if he would have waited longer than just a few minutes that radiation that the CO2 "slows" would have eventually reached his camera.
Keep it up, none of us science "deniers" can shoot down the IPCC AGW "science"
better than you can.