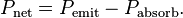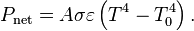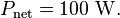Si modo
Diamond Member
Just so you know, any molecule that is IR active (meaning it has a dipole moment, which, incidentally CO2 does not, but that is another story) absorbs specific quanta of energy correlating to its functional groups with influences on the exact quanta from neighboring groups to that functional group.You're playing with words. CO2 traps energy and then re-emits it. Since statistically half would be emitted towards earth, what would that extra energy be doing, except to contribute to warming?
Once IR radiation is absorbed, it is converted into molecular vibrations (symmetric, asymmetric, in-plane, out-of-plane); the IR energy is converted to kinetic energy.
There is no trapped energy.
If there was, we would have no need for oil.
If there was, we would have no need for any electrical cells, either.
How would that mean we have no need for oil? The energy trapped by greenhouse gases is high entropy, I don't see how we can produce electricity or power of any kind with it.
Greenhouse gases do indeed cause the Earth's atmosphere to trap more energy, elsewise we'd all be frozen right now. They raise the equilibrium temperature of the Earth by making it harder for energy that enters the atmosphere to be re-radiated, requiring a higher temperature for the power flows in and out to balance one another. Its just like a blanket. If you throw a blanket over yourself, you won't get warmer and warmer without end - eventually an equilibrium is reached - but its obvious the blanket is trapping energy elsewise they'd be useless for warming you up.
CO2 does not "trap energy", as the poster stated. It's just a fact and nothing to fear.