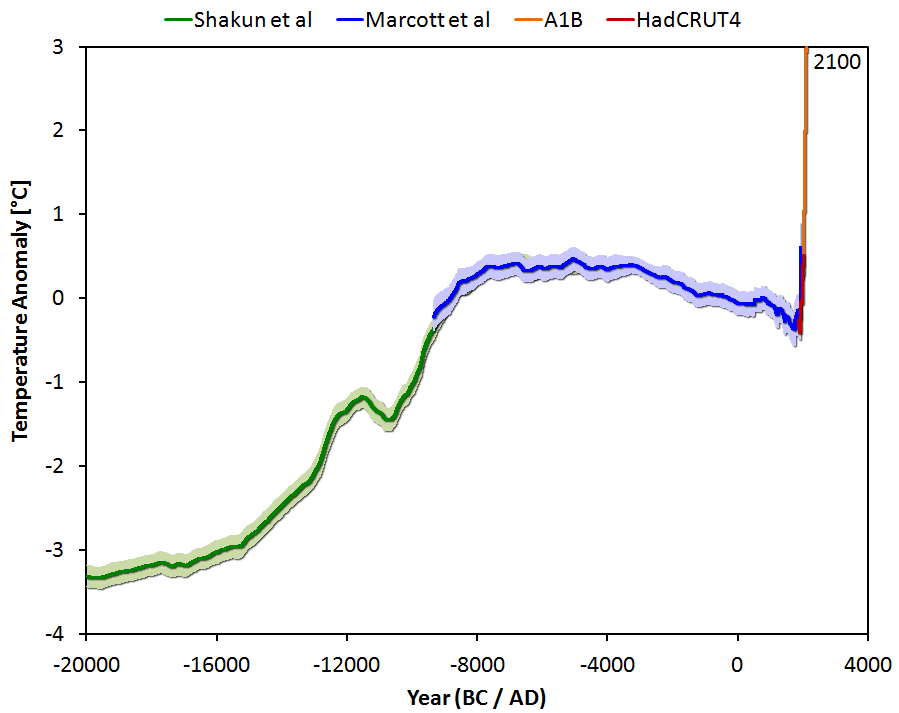
These early atmospheric scientists began this effort to model the atmosphere with the basic physics of gases and air known since the 1800's from the ideal gas law, 1st Law of Thermodynamics, Newton's second law of motion (F=ma=mg), the physical chemistry of molecular weights, partial pressures of each gas, heat capacities of individual gases and air at both constant pressure and constant volume, the gravitational acceleration constant, barometric formulae, Boltzmann's constant, Avogadro's number, mean atmospheric molecular weights, number density of individual species, total number density, atmospheric mass density, mole volume, scale height,
geopotential height of gravitational potential energy (PE), mean air-particle speed, mean free-path of air molecules, mean collision frequency, calculated speed of sound, dynamic viscosity, kinematic viscosity, coefficient of thermal conductivity, and on and on...
And
never once used any "radiative forcing" from
any IR-active greenhouse gases or
any radiative calculations from any greenhouse gases whatsoever to produce an accurate 1-D model that could calculate Earth's entire pressure, mass density, temperature, and
molecular-scale temperature as a function of geopotential altitude (
geopotential height ~ geopotential altitude ~ gravitational potential energy (PE)) profile from the surface to the edge of space.
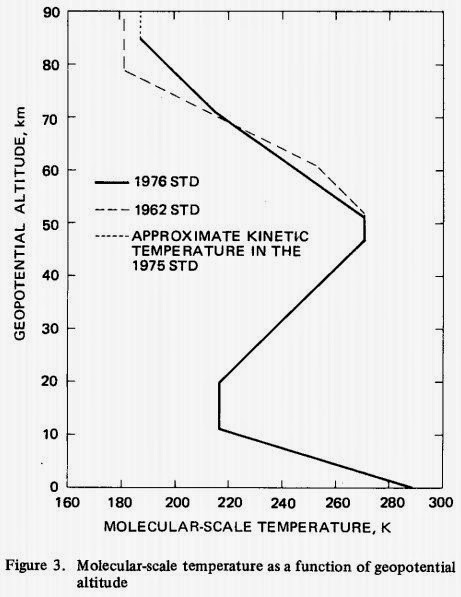
Fig. 3 from the 1976 US Standard Atmosphere document below. Note the "Molecular-scale temperature
is a function of the geopotential altitude." Thus, the kinetic temperature of the particular molecular masses and compositions of the atmosphere
is a function of the geopotential height (which is the gravitational potential energy (PE) accumulated at that height) which
adiabatically sets the
pressure at that geopotential height. This is another way of saying
temperature is a function ofatmospheric mass/gravity/pressure, which is exactly what the Maxwell atmospheric mass/gravity/pressure 33C greenhouse effect claims, not "radiative forcing" from greenhouse gases.
The entire
US Standard Atmosphere physical model was derived from physical laws assuming a completely
dry atmosphere without
any water vapor (the so called primary greenhouse gas) or
anynatural or man-made CO2 whatsoever (since CO2 at 0.03-0.04% contributes negligibly to atmospheric mass). It was only after the entire
dry atmosphere model was finished that the average water vapor in Earth's atmosphere was added back in solely by changing one
essentially constant parameter (on an annual & global basis):
the heat capacity of air at constant pressure (Cp)
(on a global annual average basis)
of the atmosphere to calculate the tropospheric lapse rate, since water vapor has a high heat capacity more than double that of N2, O2, and CO2 (which are all lower and close to the same). This is the primary means (other than clouds) by which water vapor cools the Earth surface and atmosphere, since by the lapse rate equation
dT/dh = -g/Cp
where
dT = change in temperature
dh = change in altitude
g = gravitational acceleration constant
Cp = heat capacity at constant pressure
the change in temperature dT is
inversely related to a change in heat capacity (Cp). Note also that temperature T is a function of the constants gravity (g) and (Cp), and that neither g or Cp (
within the temperature range of Earth's atmosphere) are a function of temperature (T).
Since water vapor increases heat capacity (Cp) in the lapse rate equation, it
decreases the temperature (T) at any altitude (h) including the surface where (h) = 0, and
decreases the lapse rate by half from the dry rate 9.8 C/km to the wet rate of 5 C/km if the atmosphere is fully saturated with water vapor.
The scientists involved with the US Standard Atmosphere calculated the global average lapse rate of 6.5 K/km on the basis of the known heat capacity constants of water vapor and measured global average water vapor concentrations, to calculate and confirm with observations that it is ~6.5 C/km, intermediate as expected between the dry and wet fully saturated adiabatic lapse rates, and thus this is the value used to calculate the global annual average temperature, pressure, and density profile of the Standard Atmosphere. We now know from weather balloon and satellite measurements that the global average total water vapor is quite constant, and there is little agreement about whether it has stayed the same or slightly increased or decreased over the past 35 years of the satellite era.
The tiny 0.03-0.04% of CO2 in the atmosphere does not contribute in any significant way to atmospheric molecular mass, molecular density, partial pressures, heat capacity (Cp), etc., thus in the multiple versions of the US Standard Atmosphere models, the calculated CO2 effect on the atmospheric temperature was so negligible that the atmospheric scientists
thereafter completely discarded CO2 from their model calculations of the atmosphere. The same potential effects were calculated for what is called today the "20 times stronger greenhouse gas than CO2" methane; these atmospheric scientists found the mass contribution and heat capacity to the atmosphere from methane was far too negligible to consider, thus, it was also discarded from the model along with CO2.